Prev Page--Problem, Methodology || Next Page--Site 6
Installation and calibration procedures
Observation wells
Observation wells at the sites consisted of two types: 5-cm (2-in.) water-quality monitoring wells, and 13-cm (5-in.) water-level monitoring wells. Extra precautions were taken during installation of the water-quality monitoring wells to prevent contamination through drilling procedures. A hollow-stem auger rig was used to drill all 5-cm (2-in.) wells. The augered hole diameter was 17.2 cm (6.75 in.). Clean rubber gloves were used to handle PVC casing and auger flights. Each auger flight section was washed with potable water and then acetone and rinsed with potable water (the potable water tested negative for atrazine). During augering, cuttings were removed from the edge of the augered hole. Both the PVC well screen and the casing were washed with potable water, scrubbed inside and outside with acetone, and thoroughly rinsed with potable water. When the desired well depth was reached, the screen and casing were lowered inside the hollow auger stem using thoroughly cleaned vise grips to hold the casing. The hollow-stem auger was filled with potable water to prevent sand from lodging between the casing and the stem when the stainless steel plate at the bottom of the auger was knocked out. The auger flights were then pulled out of the hole, leaving the well casing in place. The hole was gravel-packed using coarse-grained sand to 1.5 m (5 ft) above the top of the screen. From the top of the gravel pack to the surface, the hole was sealed with Volclay. Well development was achieved by bailing using a 0.9-m (3-ft) Teflon or a 1.8-m (6-ft) PVC bailer for the shallower wells or a PVC Brainard-Kilman hand pump. The bailers and hand pump were cleaned with acetone and water between sampling of different wells.
The 13-cm (5-in.) water-level observation wells were constructed using a mud rotary drilling rig. After reaching the desired depth, the mud was flushed out of the hole by pumping fresh water down through the drill stem, forcing the mud out through the annular space of the drilled 20-cm (8-in.) hole. The drilling mud was then pumped out of the mud pit. The 13-cm (5-in.) PVC screen and casing were then set to the proper depth, followed by dumping fine gravel into the water-filled hole, thus displacing the water up and out of the well. The gravel packing continued until the gravel reached approximately 1.5 in (5 ft) above the top of the screen. Bentonite pellets were then inserted around the casing on top of the gravel pack, followed by bailing a few casing volumes so as to pull the bentonite plug down on top of the gravel pack. Volclay grout was then pumped in through a tremie pipe until the annular space was filled to the ground surface. After letting the grout settle for a few days, the well was completely developed by bailing, pumping water out of the well, or air lifting.
Table 2 summarizes all the well information at the three sites. It should be noted that these sites are part of a larger network of sites monitored year-round for ground-water recharge estimation.
Table 2--Summary data on observation wells at the three study sites.
| Site | Observation well diameter |
Total depth | Screen interval | Water-level recorder |
|||
|---|---|---|---|---|---|---|---|
| cm | in. | m | ft | m | ft | ||
| 6 | 5 | 2 | 6.1 | 20 | 4.6-6.1 | 15-20 | |
| 6 | 5 | 2 | 10.1 | 33 | 8.5-10.1 | 28-33 | |
| 6 | 5 | 2 | 13.7 | 45 | 12.2-13.7 | 40-45 | |
| 6 | 13 | 5 | 16.5 | 54 | 10.4-16.5 | 34-54 | yes |
| 7 | 5 | 2 | 9.1 | 30 | 6.1-9.1 | 20-30 | |
| 7 | 5 | 2 | 15.2 | 50 | 12.2-15.2 | 40-50 | |
| 7 | 5 | 2 | 21.3 | 70 | 18.3-21.3 | 60-70 | |
| 7 | 13 | 5 | 12.0 | 39.5 | 5.9-12.0 | 19.5-39.5 | yes |
| 7 | 13 | 5 | 24.4 | 80 | 21.3-24.4 | 70-80 | yes |
| 10 | 5 | 2 | 13.7 | 45 | 10.7-13.7 | 35-45 | |
| 10 | 5 | 2 | 18.3 | 60 | 15.2-18.3 | 50-60 | |
| 10 | 13 | 5 | 16.8 | 55 | 10.7-16.8 | 35-55 | |
| 10 | 13 | 5 | 19.8 | 65 | 13.7-19.8 | 45-65 | |
| 10 | 13 | 5 | 27.4 | 90 | 24.4-27.4 | 80-90 | yes |
Neutron-probe access tubes
A 5-cm (2-in.) galvanized steel pipe was used as an access tube for the neutron probe at each site. Its bottom was sealed with a rubber stopper using adhesive sealant to prevent water from entering the pipe. The access tube installation and probe calibration were performed simultaneously, as described in what follows.
A Madeira bulk-density sampler was driven at 15-cm (6-in.) intervals into the ground. At each 15-cm (6-in.) depth interval, the sampler was pulled out and putty knives inserted into the sampler slots, thus isolating a known volume of soil [60 cm3 (3.7 in.3)] . Excess soil from both ends of the sampler were discarded, the putty knives were pulled out, and the soil sample was inserted into a plastic bag and weighed immediately so that soil-moisture loss from the sample was minimized. The 15-cm (6-in.) hole depth interval was cleaned with a 5-cm (2-in.) hand auger, and the Madeira sampler was reinserted for another 15-cm (6-in.) deep sampling. The process was repeated until the required depth was reached. For site 6 the augered depth was 320 cm (10.5 ft), and for site 7 it was 455 cm (15.0 ft). The access tube was then inserted into the 5-cm (2-in.) hole and pushed down with a twisting motion, creating a snug fit. The neutron probe was inserted into the access tube, and three 30-s field neutron readings were taken at depth intervals of 15 cm (6 in.). A set of 10 standard count neutron readings were taken before inserting the probe in the access tube.
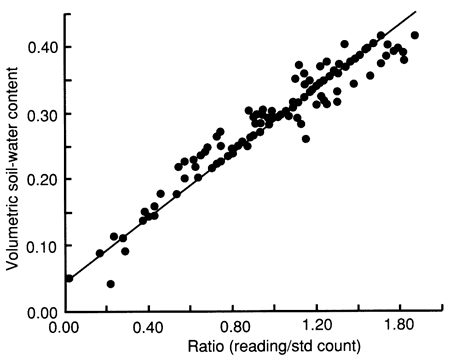
The soil samples were subsequently oven-dried, and the bulk density and moisture content on a dry weight basis were calculated, from which the volumetric water content was determined. The ratios (R's) of field to standard neutron counts for all 15-cm (6-in.) depth intervals were regressed against the corresponding measured volumetric moisture content values. A regression equation was derived to convert neutron count readings to soil-moisture content (Θ = -0.0672 + 0.2695R). A common calibration curve was developed by combining the calibration results from each site, as shown in fig. 3, to increase significantly the number of data points in the regression analysis. A linear least-squares curve, with a correlation coefficient of 0.9507, was fitted through the data.
Figure 3--Field calibration curve for neutron probe for sites 6, 7, and 10.
Tensiometers
Tensiometers were constructed in-house from 100-kPa (1-bar) ceramic cups (Soil Moisture Corporation, part 655X1-B1M1) epoxied to 1.9-cm (0.75-in.) PVC pipe. A 15-cm (6-in.) clear acrylic tube snugly fitting inside the pipe was also epoxied on top, with a rubber septum providing a sealing cap. A pressure transducer needle probe (tensimeter) inserted into the tensiometer rubber septum was used to read the capillary pressure. Before being inserted into the soil, all tensiometers were checked for leaks by filling them with water and letting them stand for 24 hr in plastic buckets with water covering the ceramic cups. After these checks the tensiometers were filled with deaired, deionized water and installed near the neutron-probe access tube as follows. A hole was dug with the 5-cm (2-in.) hand auger, down to 15 cm (6 in.) above the required depth. A hollow tube, a few millimeters smaller in diameter than the tensiometer ceramic cup, was used to core the final 15 cm (6 in.). The collected soil from the bottom of the hole was mixed with water to form a slurry, which was then poured into the hole. The tensiometer was inserted in the hole to a snug fit. The rest of the hole was then backfilled with the augered soil while water was sprayed into it. The septum was protected from exposure to weather by covering the above-ground portion of the tensiometer with a 2.5-cm (1-in.) capped PVC pipe.
Suction lysimeters
The suction lysimeters (Soil Moisture Corporation, soilwater samplers 1900 series) were first filled with deionized water to a level above the ceramic cup to wet the capillary pores. A 5-cm (2-in.) hand auger was used to auger the holes to the appropriate depth [15-180 cm (0.5-6 ft)]. A silica flour slurry was mixed with water, the hole wetted, and the slurry poured down the hole. The water was dumped out of the lysimeter, and the lysimeter was inserted into the hole with a twisting motion. The rest of the hole was grouted with a slurry made of the augered soil and water. The surface portion of the hole was filled with bentonite. A vacuum was then applied to the lysimeter.
Soil temperature thermistors
A hole was hand augered with the 5-cm (2-in.) auger to 275 cm (108 in.) below ground surface. Thermistors (YSI series 400 general-purpose probes) were taped to a 3-m (10-ft) wooden dowel at the following places: 7.5, 15, 23, 30, 45, 60, 90, 120, 150, 210, and 275 cm (3, 6, 9, 12, 18, 24, 36, 48, 60, 84, and 108 in., respectively). The thermistor dowel was then inserted into the hole. A slurry made of the cuttings and water was poured around the dowel and packed. The thermistor cables at the surface were covered with a plastic bag and further protected by covering the protruding dowel with a 15-cm (5-in.) long capped PVC casing. A YSI telethermometer was used to read the thermistor temperatures.
Chemical formulation of applied solutions
Farmers in the Great Bend region normally apply 2.24 kg/ha atrazine, or 0.22 g/m2 (2 lb/acre atrazine for corn) using ground-spray equipment, center-pivot irrigation systems, or occasionally aircraft. The atrazine is incorporated into the upper 15 cm (6 in.) of soil. One acre-foot of soil (1233 m3) weighs approximately 4 million pounds or 1814 metric tons (Richards, 1969); thus 1 acre-half-foot of soil (616 m2) weighs approximately 2 million pounds (907 metric tons). Thus the atrazine concentration usually applied by farmers is 2 lb atrazine per 2 X 106 lb soil, or 1.0 mg/kg atrazine. We increased the quantity of atrazine in the flooding experiment solution to 1.5 mg/L (which amounted to 0.45 g/m2 for site 6 and to 1.0 g/m2 for site 7) to ensure that enough atrazine was available for possible downward leaching during one field application. Liquid Atrazine 4L herbicide (Platte Chemical Co., Inc.), which contains 4 lb (1.814 kg) active ingredient per gallon (3.785 L) (or 40.8% active ingredient; that is, 479 g/L atrazine active ingredient) was used to prepare the chemical solutions. A concentration of 1.5 mg/L of atrazine equals 5.68 x 10-3 g/gal atrazine, and because the active ingredient in Atrazine 4L is 40.8%,
5.68 x 10-3 / 0.408 = 1.39 x 10-2 g/gal
Atrazine 4L was required. Because a 500-gal (1.89-m3) tank was available for mixing the chemical solution, 6.96 g Atrazine 4L was mixed in the tank filled with municipal water from Stafford (which tested negative for atrazine) to obtain the 1.5 mg/L required concentration of atrazine active ingredient.
The concentration of bromide tracer (5 meq/L or 400 mg/ L) was selected to be high enough to exceed by several times the background levels after more than a hundredfold dilution by soil solutions. The tracer solution was prepared with sodium bromide (NaBr) because sodium is the most common of the monovalent cations in the soil solution. The solution was prepared by adding NaBr to the same water to which the atrazine was added in the tank. This required 973.7 g NaBr (514.5 mg/L NaBr) to 1.89 m3 (500 gal) water. The measurement of the water volume in the tank was inexact; thus samples of the final solution were analyzed to determine the exact concentrations of both bromide and atrazine.
At site 6, 1.89 m3 (500 gal) chemical solution containing atrazine and bromide was applied after the soil profile was nearly saturated with municipal water from Stafford. At site 7, 3.79 m3 (1000 gal) chemical solution of the same concentration as that at site 6 was directly applied without presaturating the site.
Chemical analysis methodology
Atrazine was determined in soil extract solutions and water samples at the Kansas State Board of Agriculture Pesticide Laboratory using a Hewlett-Packard 5890A Gas Chromatograph with an HP5470 mass selective detector. Atrazine extraction efficiencies from soil samples were 95% to nearly 100%. Bromide was determined at the Kansas Geological Survey (KGS) by automated colorimetric methods on a Technicon Autoanalyzer. The atrazine degradation by-products were determined by a commercial analytical laboratory using gas chromatography for the biodegradation products and high-performance liquid chromatography for hydroxyatrazine determination. Additional analytical methodology is described in appendix B.
Vadose zone and Pleistocene aquifer profiles
The Great Bend Prairie is characterized by mostly sandy soils underlain by relatively thin, widespread clayey layers at shallow depth [generally 1-2 m (4-8 ft) deep] and by a shallow water table [generally 4-9 m (15-30 ft) deep].
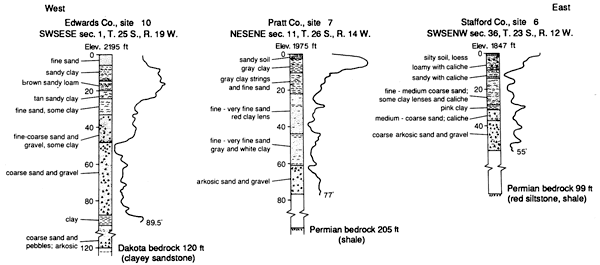
The deepest observation well at each site was gamma-ray logged. Gamma radiation generally increases as the clay content increases. Figure 4 displays the gamma-ray log traces and drill-cuttings lithology for all three study sites. A generally fining-upward sedimentary sequence can be recognized.
Figure 4--Geologic logs for each site based on drill cuttings and gamma-ray geophysical logs.
Prev Page--Problem, Methodology || Next Page--Site 6
Kansas Geological Survey, Geohydrology
Placed on web Aug. 23, 2010; originally published 1990.
Comments to webadmin@kgs.ku.edu
The URL for this page is http://www.kgs.ku.edu/Publications/Bulletins/GW12/03_inst.html