Prev Page--Geomorphology || Next Page--Well Records
Ground Water
Definition of Terms
The principles governing the occurrence of ground water have been discussed by many authors (Meinzer, 1923a; Moore and others, 1940, p. 11-32; Thomas, 1951, p. 15-34), and the reader is referred to their reports for a discussion of the subject. The following definitions of technical terms commonly used in the discussion of ground water are adapted chiefly from Meinzer (1923b).
aquifer--a rock formation, bed, or zone that contains water that is available to wells. An aquifer is sometimes referred to as a water-bearing rock or water-bearing bed.
artesian water--ground water under sufficient pressure to rise above the level at which the water-bearing rock is tapped in a well. The pressure is sometimes called artesian pressure and the rock containing artesian water is an artesian aquifer.
capillary fringe--that zone directly above the water table in which water is held in the pore spaces by capillary action.
cone of influence (or cone of depression)--a cone-shaped depression of the water table; it is developed in the vicinity of a well during pumping.
confined water--water under artesian pressure.
confining bed--generally a relatively less permeable rock layer that overlies or underlies an artesian aquifer and confines water in the aquifer under pressure.
connate water--water trapped in the openings in the rock at the time the rock was formed.
depression of the water table--lowering of the water table around a pumping well.
drawdown--depression of the water level in a well or in nearby wells during pumping.
discharge--term generally applied to ground-water discharge, the removal of water from the zone of saturation. (Meinzer, 1923b, p. 48-56).
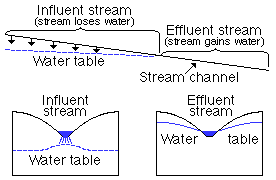
effluent stream--a stream is generally said to be effluent if water is discharged from the ground-water reservoir to the stream, sometimes called a gaining stream (Fig. 10).
Figure 10--Diagrammatic sections showing influent and effluent streams. intermittent stream-a stream that flows only at certain times following precipitation or when it receives water from springs, ground-water seepage, or melting of snow.
ephemeral stream--a stream that flows only in response to precipitation.
flowing well--an artesian well having sufficient head to discharge water above land surface.
ground water--as used in this report: water in the zone of saturation, or below the water table.
ground-water movement--the movement of ground water in or through an aquifer. The movement of ground water through rocks such as occur in the Smoky Hill Valley is extremely slow, probably less than 1 foot a day.
hydraulic gradient--gradient of the water table measured in direction of the greatest slope, generally expressed in feet per mile. The hydraulic gradient of an artesian aquifer is called the pressure gradient and is measured on the piezometric surface.
hydrologic properties--as used in this report: The properties of an aquifer that control the occurrence and movement of ground water.
hydrologic system--a series of interconnected aquifers.
hydrostatic pressure--pressure exerted by water at any point in a body of water at rest. In an artesian system the hydrostatic pressure is the artesian pressure.
impermeable rock--an impervious rock; that is, a rock through which movement of water under common subsurface pressure differentials is negligible.
infiltration--the process whereby water enters the surface soil and moves downward toward the water table.
inflow--movement of ground water into an area in response to a hydraulic gradient.
influent stream--stream that contributes water to the ground-water reservoir (Fig. 10).
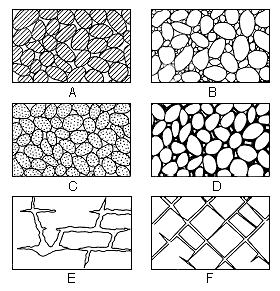
interstice--an opening or void in a rock. Interstices may be filled with air, gas, oil, water, or some other fluid. The interstices in an aquifer are filled with water (Fig. 11).
Figure 11--Diagram showing several types of rock interstices and the relation of rock texture to porosity. A, Well-sorted sedimentary deposit having high porosity; B, Poorly sorted sedimentary deposit having low porosity; C, Well-sorted sedimentary deposit consisting of pebbles that are themselves porous so that the deposit as a whole has very high porosity; D, Well-sorted sedimentary deposit whose porosity has been diminished by the deposition of mineral matter in the interstices; E, Rock rendered porous by solution; F, Rock rendered porous by fracturing. (From O. E. Meinzer.)
nonflowing artesian well--an artesian well in which the pressure is sufficient to cause the water to rise only part of the way toward the land surface.
perched water--ground water separated from the underlying water table by a zone of unsaturated rock. A perched water table is distinguished from the main water table.
perennial (or permanent) stream--a stream that flows continuously, such as Big Creek or Smoky Hill River in this report area.
permeable rock--a pervious rock, or a rock that has a texture permitting water to move through it readily under ordinary pressure differentials.
permeability--the capacity of rock to transmit water. The field coefficient of permeability of an aquifer may be expressed as the rate of flow of water at the prevailing temperature, in gallons a day, through a cross-sectional area having a thickness of 1 foot and a width of 1 mile for each foot per mile of hydraulic gradient.
piezometric surface--as generally used, the pressure-indicating surface of an artesian aquifer.
porosity--the porosity of a rock is its property of containing openings or interstices. Quantitatively, the porosity of a rock is the ratio (usually expressed as a percentage) of the volume of openings in the rock to the total volume of the rock.
recharge--the process by which water is absorbed and added to the zone of saturation. Also used to designate the quantity of water added to the ground-water reservoir.
runoff--the discharge of water through surface streams. It includes both surface-water runoff and ground-water runoff. Also used to designate the quantity of water discharged as runoff.
specific capacity--the yield of a well generally expressed in gallons a minute per foot of drawdown after a specified time of pumping.
specific yield--the specific yield of a saturated rock is the ratio of the volume of water it will yield by gravity to its own volume.
storage--water stored in openings in the zone of saturation is said to be in storage. Discharge of water not replaced by recharge from an aquifer is said to be from storage.
transmissibility--the transmissibility of a rock is its capacity to transmit water under pressure. The coefficient of transmissibility is the field coefficient of permeability multiplied by the saturated thickness, in feet, of the aquifer.
underflow or subsurface movement--underflow is the movement of ground water through a formation.
water table--the water table is the upper surface of the zone of saturation where that surface is not formed by an impermeable rock. The water table is not a plane surface, but has irregularities much like the land surface. The configuration of the water table is shown on Plate 2 by means of contours.
zone of aeration--the zone between the land surface and the water table.
zone of saturation--the zone of permeable rocks saturated with water under hydrostatic pressure.
Water-Table Conditions
Movement of Ground Water
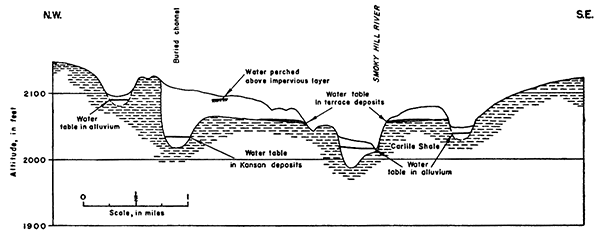
In southern Ellis County and southeastern Trego County, ground water is under water-table conditions in the Pleistocene rocks and, locally, in weathered zones in the Cretaceous rocks. The water-bearing characteristics of each of these rocks has been discussed in the section on geology. The source of ground water is precipitation on and upstream from the areas underlain by these rocks and infiltration from ephemeral streams that carry runoff from adjoining areas and cross the outcrops of these rocks. Throughout much of the upland both north and south of Smoky Hill River, the general water table is absent and small isolated bodies of ground water occur in alluvial deposits or in weathered zones in the Carlile Shale. Contours on the water table, where it can be contoured, are shown on Plate 2.
Movement of ground water is normal to the water-table contours, that is, in the direction of the greatest slope of the water table. Near Victoria the movement of ground water in the undifferentiated Pleistocene rocks is toward Big Creek and North Fork Big Creek. In the southeast part of T. 14 S., R. 17 W., the water table slopes toward Big Creek at about 17 feet per mile and just south of Victoria the slope toward North Fork Big Creek is about 25 feet per mile. Movement of ground water in the alluvium of Big Creek is downstream and the slope of the water table is about 8 feet per mile. In the alluvium along North Fork Big Creek the downstream slope of the water table is about 9 feet per mile. Big Creek and North Fork Big Creek are effluent streams and gain water from ground-water discharge in this area.
In southwestern Ellis County, the slope of the water table in the Crete Formation is about 22 feet per mile and ground-water movement is toward the river and downstream. In the alluvium of Smoky Hill River the slope of the water table is about 9 feet per mile in Trego County and about 7 feet per mile in southeastern Ellis County. Movement of ground water in the alluvium is principally downstream, but along the valley margins some movement is toward the center of the valley. Smoky Hill River is a gaining stream throughout its course in the report area (Pl. 2).
Interrelation of Precipitation, Runoff, and Recharge
In central Kansas the original source of all ground water is local precipitation. Part of the precipitation evaporates directly from the land surface on which it falls; part seeps into the soil and is absorbed by the roots of plants and transpired; part runs off in surface streams; and a part infiltrates the ground and percolates downward to the ground-water reservoir. Evaporation takes place whenever moisture is available and humidity, temperature, and air movement are suitable. Usually, air movement is adequate and humidity is low, hence moisture will evaporate whenever it is available at or near the land surface. During the growing season, plants transpire moisture whenever it is available in the soil or whenever the water table is shallow enough to be reached by the roots. During the precipitation, water seeps into the soil until the capacity of the soil to absorb water is satisfied, after which the excess runs off in surface streams. When precipitation falls at a rapid rate, a large proportion of the precipitation becomes runoff, and streams may Hood their valleys. The amount of water that runs off a given area is measured by gaging the streams that drain the area. In Table 2 runoff is expressed in inches. Runoff is 1.00 inch when the amounts of water flowing from a drainage area would form a layer 1.00 inch deep if spread uniformly over the area.
Table 2--Discharge of Smoky Hill River near Ellis and Russell. Records collected by Surface Water Branch and published in Water-Supply Papers, U. S. Geological Survey.
| Year | Annual discharge, acre-feet |
Increment, Ellis to Russell, acre-feet |
Runoff, Ellis to Russell, inches |
Precipitation at Hays, inches |
Percent of precipitation that is runoff |
|
|---|---|---|---|---|---|---|
| Near Ellis |
Near Russell |
|||||
| 1942 | 78,110 | 191,200 | 113,090 | 1.59 | 29.61 | 5.4 |
| 1943 | 12,300 | 21,400 | 9,100 | .13 | 16.19 | .8 |
| 1944 | 67,640 | 191,300 | 123.660 | 1.74 | 29.70 | 5.9 |
| 1945 | 12,000 | 65,830 | 53,830 | .76 | 20.34 | 3.7 |
| 194G | 113,500 | 175.000 | 61,500 | .86 | 26.48 | 3.3 |
| 1947 | 32,710 | 97,250 | 64,540 | .90 | 22.65 | 4.0 |
| 1948. | 37,350 | 72,370 | 35,020 | .49 | 26.19 | 1.9 |
| 1949 | 129,400 | 228,100 | 98.700 | 1.39 | 23.62 | 5.9 |
| Average | 0.98 | 3.9 | ||||
The amount of water that runs off the area of this report is approximately the difference in the How of Smoky Hill River between the Ellis and Russell gaging stations. Between these two stations, surface drainage flows into Smoky Hill River from the valley, valley slopes, and the drainage basins of Big Creek and Big Timber Creek. The area drained between these two stations is 1,335 square miles and includes all the area covered by this report. Table 2 gives the annual discharge in acre-feet of Smoky Hill River at the gaging stations near Ellis and near Russell, the increase in runoff between the two stations, the runoff in inches from the drainage area between the two stations, and the ratio of runoff to the precipitation at Hays for the period 1942-1949. During this period runoff ranged from 0.13 inch in 1943, when precipitation at Hays was 16.19 inches, to 1.74 inches in 1944, when precipitation was 29.70 inches. In general, runoff is greatest when precipitation is above normal and least when precipitation is below normal. The average runoff during this period was about 1 inch, or approximately 4 percent of the precipitation.
The amount of water available to recharge the ground-water reservoir is the amount in excess of that discharged by evaporation and transpiration and by surface runoff into the streams. Recharge takes place by infiltration of water from the soil, when it is saturated, downward to the water table; by the infiltration of water into deep cracks and other openings that extend below the zone from which plant roots obtain water that is transpired; and by seepage of water into the ground from streams that are above the water table. A large part of the recharge in the Smoky Hill Valley probably comes from precipitation falling on the nearly Hat terrace surface where conditions are more favorable for infiltration than for runoff. Additional recharge is received from ephemeral streams that carry runoff across the terrace surface and have channels above the water table. Water seeps into the ground along the banks and beds of these streams and percolates to the water table. Normally, Smoky Hill River and Big Creek are lower than the water table and receive water from the ground-water reservoir, but during Hood stages the streams may contribute a small amount of recharge to the zone of saturation by lateral seepage into the channel banks and by vertical infiltration into the alluvial material they overflow.
The hydrographs of several observation wells (Fig. 12) show the relation between precipitation and fluctuations of the water table. Wells 14-16-17cb and 15-18-1bb show that the water table rose in the summers of 1947 and 1949 following periods of heavy rainfall and declined in the fall of 1947 and 1948 following periods of light rainfall. The water table reached a high point several weeks after the period of excessive precipitation. The time interval represents the time required for percolation of ground water to the water table.
Figure 12--Hydrographs of five observation wells.
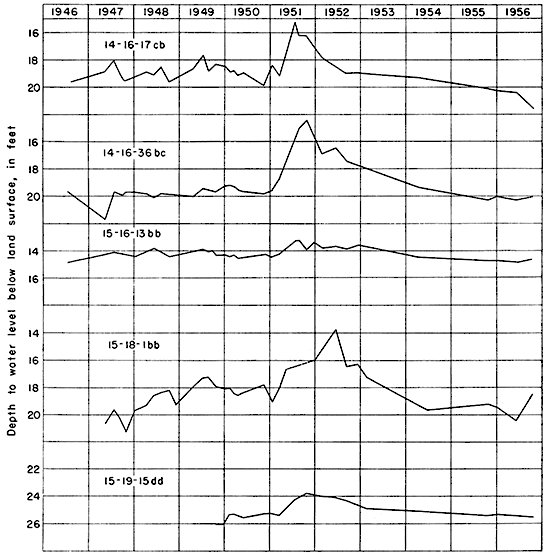
An approximation of the amount of recharge can be obtained by multiplying the magnitude of the rise of the water table by the specific yield of the water-bearing material. If the specific yield is assumed to be 15 percent, then the rise of the water level of well 14-16-17cb of 0.8 foot in June and July, 1947, and of 1.0 foot in May and June, 1949, indicates recharge in the vicinity of this well to have been 0.12 foot in June and July, 1947, and 0.15 foot in May and June, 1949. The effect of this recharge seemingly was offset by increased discharge of ground water into the channel of North Fork Big Creek. Generally, recharge is greater during periods of above-normal precipitation and less during periods of below-normal precipitation. Probably the average annual recharge over a long period of time is about 0.3 to 1 inch a year.
Fluctuations of the Water Table
The surface of the ground-water body is not stationary but fluctuates much like the water level in a lake or reservoir. Over a long period of time under natural conditions, a state of approximate equilibrium is attained between the amount of water added annually to ground-water storage and the amount discharged annually. The water table rises when recharge exceeds discharge and declines when discharge exceeds recharge.
In 1947, 14 wells were selected for observation wells, and the water levels in some of these wells have been measured periodically since that time. The descriptions of the wells and the measurements of water levels in them have been published in the annual waterlevel reports of the U. S. Geological Survey for the years 1947 through 1955. The measurements for 1956, 1957, 1958, and 1959 were published in Kansas Geological Survey Bulletins 125, 131, 141, and 146 and measurements for ensuing years will be published as bulletins. Figure 12 is a graphic representation of the changes in water levels in five of these wells. Precipitation and cumulative departure from normal precipitation at Hays are shown in Figure 3. A comparison of the hydrographs in Figure 12 with the precipitation records in Figure 3 shows a correlation between rise in water levels and periods of excessive precipitation, and decline in the water levels and periods of subnormal precipitation. The water levels in wells 14-16-17cb and 14-16-36bc rose about 4 and 5 feet, respectively, during the first half of 1951, owing to the heavy precipitation that caused the floods of May, June, and July, 1951. During the period from August 1946 to February 1952 the net rise in water level in well 14-16-17cb was 1.8 feet, in well 14-16-36bc was 2.8 feet, and in well 15-16-13bb was 1 foot. During this period, the net cumulative departure above the normal precipitation was 32.47 inches. During the period from February 1952 to October 1956 the net decline in water level in well 14-16-17cb was 3.8 feet, in well 14-16-36bc was 3.1 feet, and in well 15-16-36bb was 0.9 foot. During this period the net cumulative departure below the normal precipitation was 31.11 inches. During the period August 1946 to October 1956 water levels in wells 14-16-17cb and 14-16-36bc had a net decline of 2 feet and 0.3 foot, respectively, and in well 15-16-13bb a net rise of 0.1 foot. During the period the net cumulative departure below normal precipitation was 0.64 inch.
In general, the water table is high during wet periods and is low during dry periods. During wet seasons, more water is added to ground-water storage because recharge is greater, and more water is available in the soil for transpiration and evaporation. During dry seasons, more ground water is transpired by plants whose roots tap the water table; streams are so low that ground water is discharged into them; and recharge is at a minimum. Thus, groundwater levels tend to decline during periods of drought. In general, the fluctuations of the water table are approximately cyclic, just as are seasons of wet and dry weather.
The Ground-Water Reservoir
The main water table--In the Smoky Hill Valley the main ground-water reservoir is in the alluvial materials adjacent to Smoky Hill River. In southwestern Ellis County and southeastern Trego County the alluvium is separated from the older Pleistocene deposits by shale walls that confine the ground-water reservoir to the narrow trough of the inner valley. The water table in the alluvium is higher than Smoky Hill River and ground water is discharged into the river except during flood stages. The water table in the alluvium of the valleys of the principal tributaries to the river is continuous with the water table in Smoky Hill Valley, and ground water moves down these valleys into Smoky Hill Valley. The depth to water in the alluvium of Smoky Hill Valley may be as much as 21 feet, but is less than 15 feet in many places. At the present time, the alluvium is tapped by only a few wells, but it is potentially the source of much greater supplies in this area. One of the Hays municipal wells has been test-pumped at a rate of 860 gallons a minute, and wells of large yield probably could be constructed at any place in the valley where the saturated part of the alluvium is thick.
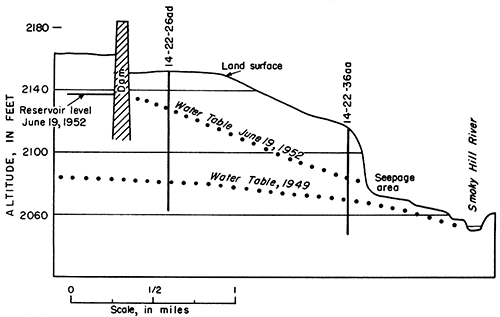
In southeastern Trego County the main water table extends into the narrow meandering bedrock channel filled with deposits of Kansan age (Fig. 7, Pl. 2). Water from the alluvium moves down- stream through this channel and back into the alluvium again where the channel joins the alluvium. In 1949 the depth to water in the Kansan deposits was 71.86 feet in well 14-22-26ad and 42.65 feet in well 14-22-36aa. The maximum thickness of saturated material was about 32 feet. In June 1952 the water level in well 14-22-26ad was 25.49 feet below land surface, and in well 14-22-36aa was 30.52 feet below land surface. Figure 13 shows the relation of the water table in the Kansan deposits to the reservoir and river near the Cedar Bluff dam in 1949 and in 1952.
Figure 13--Water table in Pleistocene rocks near Cedar Bluff dam in 1949 and 1952.
In southeastern Ellis County the main water table in the alluvium is continuous with the water table in the Crete deposits underlying the Pfeifer Terrace. In this area the depth to water ranges from 14 to 35 feet, being greatest in the terrace areas and least in the alluvium next to the river.
Along Big Creek the main water body extends through the undifferentiated Pleistocene deposits that underlie the broad high terrace north of the stream. The water table in these deposits is higher than the creek, which gains water from the ground-water reservoir along its course. In this area the depth to water ranges from 12 feet to 55 feet (Pl. 2). In the alluvium adjacent to Big Creek and North Fork Big Creek the depth to water is generally less than 20 feet. The depth to water is greatest in the uplands, the maximum depth being along the divide between Big Creek and North Fork Big Creek.
Along part of its course in Ranges 16 W. and 17 W., the south bank of Big Creek is formed by walls of relatively impervious shale in the Greenhorn Limestone. The shale beds isolate small bodies of ground water in the upland from the alluvium near the creek, but almost no ground water moves toward the creek from the shale outcrop areas.
Isolated water bodies--Much of the ground water available to wells in the area of this report is in small ground-water bodies not connected to the main bodies along the principal streams. These water bodies occur in the Crete deposits under the Pfeifer Terrace, in the Nebraskan(?) deposits in the Antonino ridge, and in the alluvium of the upland. Figure 14 illustrates water tables in various ground-water reservoirs in southeastern Trego County.
Figure 14--Water tables in Pleistocene rocks in southeastern Trego County. water table in the Crete Formation has been contoured. Near Cedar Bluff dam the water table in the Crete deposits is 30 to 40 feet above the water table in the nearby alluvium, but the difference in altitude of the two water tables decreases downstream, and at Schoenchen they are only 20 feet apart.
Along much of the valley west of Schoenchen and locally in southeastern Ellis County, the terrace deposits of the Crete Formation are separated from the alluvium by shale bluffs, which form boundaries between the main water table adjacent to the river and the body of ground water in the Crete Formation. On Plate 2 the Along the shale bluffs between the Pfeifer Terrace and Smoky Hill River Valley, locally along the upland margin of the terrace, and where tributary streams have cut through the Crete deposits and intrenched the underlying shale are small areas where the Crete deposits are dry. Seepage occurs locally along the shale bluffs during periods when the water table is high, and permanent springs flow where some of the major tributaries intersect the water table in the Crete Formation. In T. 15 S., R. 19 and 20 W., the channel cut into Kansan deposits is not deep enough for the water table in them to connect with the water table in the alluvium of Smoky Hill Valley. In this part of the area, the water body in the Crete Formation extends into the underlying Kansan deposits. Several permanent springs or zones of seepage mark the intersection of major tributary streams with this channel (Fig. 7).
Locally, clay lenses in the upper part of the upper Pleistocene deposits cause small zones of perched water several feet above the general water table in these deposits. Generally, these zones are wet-weather phenomena, and wells drilled into them go dry during dry seasons.
In general, the depth to water in the Crete deposits is about 17 to 53 feet and averages about 32 feet. Locally, along the margin of the terrace where springs occur, the depth to water is less than 10 feet, and several shallow wells have been constructed in such favored locations.
A small body of ground water occurs in the Nebraskan(?) deposits under the narrow ridge extending southeastward through Antonino. These deposits are remnants of a channel fill overlying the eroded surface of the Carlile Shale. They are about 5 to 20 feet thick, and the depth to water on the ridge is generally between 10 and 15 feet. No wells penetrating these deposits are reported to yield large supplies, but the village of Antonino is supplied with water from several wells of small yield.
Locally, the alluvium along small streams in the upland contains small bodies of ground water, which supply many farm wells of small yield. Ground water moves down valley in the alluvium and eventually joins the main ground-water reservoir along the principal streams. The depth to water in the upland valleys is generally less than 20 feet and the thickness of saturated material may be as much as 20 feet.
The alluvial deposits near Smoky Hill School in the southwestern part of T. 15 S., R. 19 W., contain a body of water that extends under an area of several square miles and supplies water to several farm wells. The depth to water in these deposits is between 20 and 30 feet. The direction of ground-water movement is toward the northeast, and the gradient of the water table is about 20 feet per mile (Pl. 2).
Discharge
Natural discharge--Before any wells were drilled, the groundwater reservoir in this area was in a state of approximate equilibrium; that is, the average annual recharge was balanced by the average annual discharge and the water table was moderately stable, except for fluctuations caused by seasons of excessive or abnormally low precipitation. Ground water was discharged principally by seepage into Smoky Hill River and its tributaries and by subsurface movement to the east. Small amounts of ground water are discharged by transpiration and by evaporation where the water table is shallow.
The principal methods by which ground water is discharged at the present time are by subsurface movement to the east in the alluvium, Crete deposits, and undifferentiated Pleistocene deposits along Big Creek, and by effluent seepage into Smoky Hill River, Big Creek, and other streams. Each of the principal streams in the area gains water from the ground-water reservoir (Pl. 2), and seepage is an important factor in maintaining the flow of the perennial streams.
Part of the ground water in the alluvial rocks in Smoky Hill and Big Creek Valleys moves into the area from the west by lateral seepage in the subsurface. Likewise, part of the ground water leaves the area by lateral seepage to the east.
Some water is discharged from the ground-water reservoir through transpiration hom plants whose roots penetrate the capillary fringe. Trees that line the banks of Big Creek along most of its course discharge a considerable amount of water during the growing season. Along Smoky Hill River, trees are less numerous and the discharge of ground water by transpiration is not so great. Locally, along Smoky Hill River and in some of the small upland valleys, deep-rooted crops such as alfalfa discharge ground water by transpiration, but because the areas of these crops are small, the amount of ground water transpired is not great.
Discharge from wells--In 1951, there were no irrigation or industrial wells in the area of this report, and except for the Hays municipal wells near Schoenchen, no large quantities of ground water were discharged through wells in the Smoky Hill Valley. Hays had five municipal wells that tap alluvial deposits of Smoky Hill River. According to officials of the city of Hays, about 185 million gallons of water was pumped from these wells during 1950 and about 195 million gallons in 1951. The maximum daily amount used by the city was about 1.8 million gallons in 1950 and 2 million gallons in 1951. The amount of water pumped by the city probably was more than the total amount of water pumped by all other users in the report area.
Artesian Conditions
Occurrence of Artesian Water
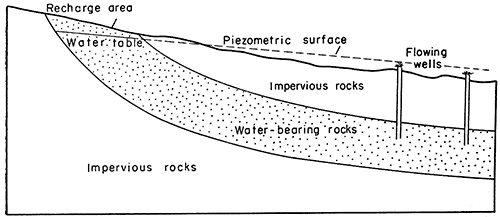
Ground water in a permeable bed confined under pressure between two less permeable beds is regarded as artesian or confined water. When a well is drilled into the permeable water-bearing formation, water will rise in the well above the level at which it is first tapped. A flowing artesian well is one in which the water rises above the land surface. In the following discussion the term artesian is used for both nonflowing and flowing wells. In the classic artesian system the permeable bed dips from an outcrop area where recharge occurs toward the area of artesian flow (Fig. 15). Where the land surface is lower than the piezometric surface of the water in an artesian aquifer, flowing wells may be constructed.
Figure 15--Diagrammatic section showing simple artesian system.
In this area wells drilled into the Dakota Formation are artesian but do not flow. Twenty-seven of the wells shown on Plate 2 obtain water from the Dakota Formation in three general areas: (1) along the upland between Smoky Hill and Big Creek Valleys in southeastern Ellis County; (2) north of the Pfeifer Terrace west of Antonino, and (3) west of the Smoky Hill School in southwestern Ellis County. Water from wells in the Dakota Formation differs considerably in quality from place to place and the artesian head seems to differ to some extent, indicating that the sandstones penetrated by the wells are not necessarily interconnected.
Recharge of Artesian Aquifers
Several investigators have postulated different ideas concerning recharge to the Dakota artesian aquifers. Russell (1928) suggested that all water in sandstones of the Dakota is connate and that the formation receives no recharge. Others (Darton, 1909a, 1909b) have regarded the Dakota as a classic artesian system having elevated intake areas along the eastern front of the Black Hills and Rocky Mountains. Latta (1941, p. 40, pl. 5) indicated that the Dakota receives recharge in outcrop areas in southeastern Colorado.
If the Dakota aquifers received no replenishment, wells tapping them would record gradually diminishing hydrostatic head and the water supply would eventually be exhausted. Wells tapping the Dakota in central Kansas are known to produce water for many years without noticeable change in head. The water levels in wells that have been measured at different times show fluctuations of the piezometric surface that can be explained only by the addition of water to the artesian reservoirs. For instance, the piezometric surface in well 15-17-19ab was 1.1 feet higher in February 1950 than it had been in July 1946. Also, Frye and Brazil (1943, fig. 7) have shown that water was moving eastward in the upper sandstone of the Dakota in Russell County. This movement could occur only if water was being added to the aquifer.
Because the sandstone beds are lenticular and discontinuous, ground water could not move readily through the Dakota for the great distances from southeastern or central Colorado to central Kansas. Therefore, a recharge area near to the area of this report seems most likely. Sandstones of the Dakota are overlain by saturated Neogene rocks in a broad area less than 100 miles south and southwest of this area (Waite, 1942, pl. 5; McLaughlin, 1949, pl. 3). Those areas are relatively near the area of this report and the Dakota might receive recharge there. Another possibility is by recharge in the report area, or nearby, by the downward percolation of water through joints and fractures in the Cretaceous shale and chalk beds or by slow percolation through the shale, which is not wholly impervious. Small seeps issuing from joints in the shale and chalk have been noted, and if the joints extended deep enough, water could move through them to the sandstones of the Dakota.
Any explanation of the method of recharge to sandstones of the Dakota must account for the differences in hydrostatic head between different sandstone bodies and for the considerable variation in chemical quality from place to place. The ground water probably is replenished in a combination of ways and from a combination of sources. The water being added to the aquifer may be mixed with connate water or may dissolve mineral matter from local soluble rock material. The entire hydraulic system in the Dakota sandstones from the recharge area through conduits to the reservoirs tapped by wells is undoubtedly extremely complex.
Utilization of Ground Water
During the course of the investigation, information was obtained on 141 wells, of which 132 are domestic or stock wells, 6 are public-supply wells, 2 are industrial wells, and 1 is a municipal test well. The principal uses of ground water in this area are for domestic,. stock, and public supplies.
Domestic and Stock Supplies
Water for domestic use is obtained almost entirely from wells, but in parts of the area a few cisterns are used. Domestic wells supply homes with water for drinking, cooking, and washing, and supply those schools not served by municipal wells. Stock wells supply water for livestock, principally cattle. Most of the water for livestock also is obtained from wells, but in recent years stock ponds have been constructed on dry water courses in areas where supplies of ground water are difficult to obtain. Although many wells obtain water from either the alluvium or terrace deposits at relatively shallow depths, some wells obtain water from the Dakota Formation at depths of as much as 200 feet.
Public Water Supplies
Hayes and Victoria are the only cities in the vicinity that have public water-supply systems. Brief descriptions of these systems are given below, and details of well construction are given in Table 11 at the end of this report.
Hays--The city of Hays, on the terrace of Big Creek, obtains its water supply from wells in the valleys of Big Creek and Smoky Hill River. Prior to the summer of 1949, the water supply for Hays was obtained from 15 wells tapping the alluvium and terrace deposits of Big Creek. Owing to a need for additional water, the city in June 1949 constructed five gravel-packed wells in Smoky Hill Valley south of Hays. These wells obtain water from sand and gravel of terrace deposits that directly underlie the flood plain of Smoky Hill River. During 1950 and 1951 approximately 50 percent of the water supply for Hays came from the five wells in Smoky Hill Valley; locations are shown on Plate 2, and additional construction data are given in Tables 3 and 11.
In 1949 Hays had 2,105 water customers each using an average of 508 gallons a day. The city has a water storage capacity of 1,100,000 gallons. Consumption was 390,566,040 gallons of water during 1949, declined to 368,658,000 gallons in 1950, and rose again to 391,371,000 gallons in 1951. The water is chlorinated, aerated to remove the iron, and softened by use of lime and soda ash. Maximum daily consumption in 1951 was about 2 million gallons.
Victoria--The city of Victoria, along North Fork Big Creek, obtains its water supply from five wells tapping the alluvium of tributaries of North Fork Big Creek. Data regarding these wells are given by Latta (1948, p. 153).
In 1949 Victoria had 266 water customers each using an average of 124 gallons a day. Victoria has a water storage capacity of 50,000 gallons and consumes about 11,650,000 gallons a year. In 1951 the only water treatment was chlorination.
Table 3--Data on Hays municipal wells in Smoky Hill Valley
| U.S.G.S. no. |
City no. |
Static water level, feet |
Depth, feet |
Length of screen, feet |
Length of casing, feet |
Yield, gpm |
Drawdown, feet |
Length of pumping test, hours |
|---|---|---|---|---|---|---|---|---|
| 15-18-28da1 | S-1 | 12.0 | 57 | 28 | 30 | 400 | 12.0 | |
| 450 | 14.0 | |||||||
| 500 | 16.5 | 72 | ||||||
| 550 | 18.3 | |||||||
| 600 | 22.0 | |||||||
| 650 | 25.0 | |||||||
| 700 | 27.0 | |||||||
| 15-18-28da2 | S-2 | 13.0 | 53 | 28 | 24 | 460 | 11.2 | |
| 600 | 13.9 | |||||||
| 800 | 20.6 | 24 | ||||||
| 830 | 21.0 | |||||||
| 860 | 24.0 | |||||||
| 600 | 15.0 | |||||||
| 15-18-27cb1 | S-3 | 11.5 | 51 | 28 | 24 | 250 | 16.0 | |
| 280 | 17.4 | |||||||
| 300 | 18.0 | 48 | ||||||
| 350 | 19.7 | |||||||
| 400 | 23.0 | |||||||
| 450 | 26.5 | |||||||
| 460 | 28.0 | |||||||
| 15-18-27cb2 | S-4 | 10.2 | 55 | 28 | 28 | 408 | 6.0 | |
| 589 | 11.0 | |||||||
| 672 | 16.0 | |||||||
| 760 | 21.5 | 48 | ||||||
| 805 | 23.0 | |||||||
| 844 | 26.0 | |||||||
| 15-18-27ca | S-5 | 11.0 | 54 | 36 | 20 | 300 | 12.5 | |
| 400 | 16.0 | |||||||
| 500 | 18.5 | 48 | ||||||
| 560 | 28.0 | |||||||
| 590 | 30.0 |
Table 4--Analyses of water from wells in southern Ellis County and parts of Trego and Rush Counties. Dissolved constituents given in parts per milliona.
| Well Number |
Depth of well below land surface, feet |
Geological source |
Date of collection |
Temperature (°F) |
Specific conductance (micromhos at 25°C) |
Silica (SiO2) |
Iron (Fe) |
Calcium (Ca) |
Magnesium (Mg) |
Sodium and potassium (Na+K) |
Bicarbonate (HCO3) |
Sulfate (So4) |
Chloride (Cl) |
Fluoride (F) |
Nitrate (NO3) |
Dissolved solids (residue on evaporation at 180°C) |
Hardness as CaCO3 |
|
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Calcium, magnesium |
Noncarbonate | |||||||||||||||||
| 14-16-10dd | 17.6 | Pleistocene undifferentiated |
3-24-1947 | 51 | 599 | 0.10b | 90 | 9.8 | 18 | 272 | 24 | 21 | 0.6 | 30 | 361 | 265 | 42 | |
| 14-16-16ab | 24.7 | (?) Greenhorn Limestone |
10-31-1949 | 56 | 934 | 32 | .34 | 150 | 8.0 | 37 | 288 | 132 | 62 | .2 | 32 | 610 | 408 | 172 |
| 14-16-17cb | 24.0 | Terrace deposits | 3-24-1947 | 53 | 631 | .05b | 103 | 9.2 | 9.0 | 282 | 38 | 28 | .3 | 3.0 | 383 | 295 | 64 | |
| 14-16-18bb2 | 34 | Terrace deposits | 11-15-1946 | 28 | .03 | 117 | 7.4 | 5.8 | 344 | 24 | 11 | .1 | 15 | 397 | 322 | 40 | ||
| c14-16-2Odd | 48.9 | Pleistocene undifferentiated |
3-24-1947 | 58 | 584 | .10b | 97 | 8.7 | 4.0 | 296 | 21 | 13 | .2 | 4.0 | 342 | 278 | 35 | |
| 14-17-17bb | 35.8 | Pleistocene undifferentiated |
3-21-1947 | 60 | 535 | .00b | 92 | 11 | 1.5 | 236 | 53 | 16 | .2 | 8.0 | 340 | 275 | 81 | |
| 14-17-25aa | 28.0 | Pleistocene undifferentiated |
3-24-1947 | 56 | 3.160 | .10b | 478 | 48 | 72 | 219 | 51 | 930 | .1 | 3.0 | 1,690 | 1,390 | 1,210 | |
| 14-18-12bb | 31.0 | Pleistocene undifferentiated |
3-21-1947 | 60 | 2,810 | .00b | 10 | 6.3 | 599 | 487 | 247 | 480 | 7.0 | 5.0 | 1,590 | 53 | 0 | |
| 14-19-29dd | 350 | Dakota Formation | 10-28-1949 | 59 | 3.840 | 20 | 3.9 | 17 | 9.6 | 845 | 540 | 268 | 836 | 6.0 | 3.9 | 2,270 | 82 | 0 |
| 14-20-28ad | 22.8 | Carlile Shale or alluvium |
10-29-1949 | 57 | 691 | 32 | .31 | 109 | 17 | 10 | 308 | 32 | 29 | .1 | 46 | 430 | 342 | 89 |
| 14-20-32aa1 | 39.4 | Crete Formation | 10-22-1949 | 57 | 634 | 35 | .14 | 86 | 14 | 29 | 264 | 40 | 30 | .2 | 42 | 422 | 272 | 56 |
| 14-20-32dd2 | 43.0 | Crete Formation | 3-21-1947 | 62 | 620 | .00b | 100 | 12 | 4.5 | 276 | 30 | 35 | .1 | 2.0 | 391 | 299 | 110 | |
| 14-20-35dc | 19.5 | Crete Formation | 3-21-1947 | 62 | 649 | .00b | 108 | 9.2 | 1.0 | 296 | 26 | 18 | .4 | 10 | 427 | 307 | 64 | |
| 14-21-30ca | 29.2 | Alluvium | 10-29-1949 | 59 | 1,040 | 32 | 4.7 | 156 | 29 | 39 | 284 | 262 | 29 | .7 | 55 | 772 | 508 | 275 |
| 14-21-31cd | 13.2 | Alluvium | 10-29-1949 | 62 | 1,210 | 39 | .10 | 174 | 43 | 43 | 268 | 416 | 23 | .6 | 22 | 938 | 611 | 391 |
| 14-21-34ba | 20.9 | Alluvium | 10-29-1949 | 58 | 1,980 | 36 | .26 | 342 | 48 | 20 | 194 | 496 | 97 | .4 | 348 | 1,480 | 1.050 | 891 |
| 14-21-35ca | 19.1 | Crete Formation | 10-22-1949 | 59 | 1,600 | 27 | .26 | 214 | 38 | 101 | 224 | 538 | 78 | .5 | 69 | 1.180 | 690 | 506 |
| 14-21-35cd | 19.8 | Terrace deposits | 10-22-1949 | 59 | 1,670 | 45 | .77 | 226 | 78 | 81 | 374 | 648 | 41 | .3 | 26 | 1,330 | 884 | 577 |
| 14-22-26ad | 90.7 | Grand Island Formation |
10-29-1949 | 510 | 35 | .19 | 76 | 11 | 20 | 248 | 53 | 9.0 | .4 | 9.0 | 368 | 235 | 32 | |
| 15-16-6cb | 56.9 | Pleistocene undifferentiated. |
10-29-1949 | 58 | 465 | 38 | .39 | 86 | 4.8 | 4.8 | 268 | 7.2 | 8.0 | .1 | 6.7 | 298 | 234 | 14 |
| 15-16-9aa | 132.6 | Dakota Formation | 3-24-1947 | 57 | 8,440 | .05b | 56 | 50 | 1,760 | 190 | 523 | 2,460 | 2.4 | 3.5 | 4.950 | 345 | 189 | |
| 15-16-14ab | 203.8 | Dakota Formation | 3-24-1947 | 55 | 929 | .10b | 121 | 14 | 47 | 311 | 106 | 44 | .7 | 40 | 571 | 359 | 104 | |
| 15-16-18bb | 188.1 | Dakota Formation | 3-24-1947 | 58 | 4,430 | .05b | 92 | 25 | 858 | 359 | 411 | 1,030 | 3.2 | 20 | 2.610 | 332 | 38 | |
| 15-16-21bd | 18.3 | Nebraskan(!) deposits |
10-29-1949 | 59 | 1,120 | 28 | .19 | 88 | 15 | 147 | 340 | 140 | 91 | 1.0 | 55 | 730 | 281 | 2 |
| 15-16-25bb | 19.4 | Crete Formation | 10-29-1949 | 59 | 635 | 31 | .04 | 96 | 6.8 | 17 | 268 | 16 | 28 | .2 | 36 | 416 | 268 | 48 |
| 15-17-5ba | 149.9 | Dakota Formation | 3-24-1947 | 58 | 6,800 | .10b | 43 | 26 | 1,460 | 382 | 461 | 1,830 | 4.0 | 5.0 | 4,020 | 214 | 0 | |
| 15-17-14dd | 170.6 | Dakota Formation | 3-24-1947 | 57 | 5,590 | .00b | 87 | 35 | 1,120 | 460 | 556 | 1,300 | .2 | 8.0 | 3,330 | 361 | 0 | |
| 15-17-25bc | 15.2 | Crete Formation | 3-24-1947 | 50 | 501 | .05b | 90 | 5.5 | 5.2 | 232 | 29 | 6.5 | .3 | 35 | 332 | 247 | 57 | |
| 15-17-33dc | 16.7 | Terrace deposits | 10-29-1949 | 57 | 1,590 | 34 | .24 | 258 | 38 | 9.4 | 326 | 632 | 55 | .2 | 2.4 | 1,190 | 800 | 533 |
| 15-18-1bb | 32.8 | (?)Carlile Shale | 10-31-1949 | 56 | 3,100 | 29 | 9.5 | 627 | 68 | 148 | 396 | 1,570 | 144 | 1.0 | 1.7 | 2,780 | 1,840 | 1,520 |
| 15-18-9bb | 269.5 | Dakota Formation | 10-31-1949 | 59 | 3,640 | 38 | 562 | 89 | 341 | 280 | 1,780 | 298 | 1.0 | 4.1 | 3,250 | 1,770 | 1,540 | |
| 15-18-13ab | 26.7 | (?)Carlile Shale | 3-24-1947 | 54 | 2,760 | .10b | 662 | 19 | 13 | 298 | 1.260 | 101 | 1.3 | 70 | 2,280 | 1,730 | 1,490 | |
| 15-18-16bb | 16.4 | Alluvium | 3-21-1947 | 51 | 2,080 | .00b | 403 | 30 | 58 | 282 | 844 | 137 | .5 | 20 | 1,660 | 1,130 | 899 | |
| 15-18-20ab | 23.5 | Alluvium | 10-31-1949 | 59 | 6,090 | 32 | 479 | 49 | 767 | 412 | 84 | 1,870 | .1 | .8 | 3,500 | 1,400 | 1,060 | |
| 15-18-22bb | 47.2 | Nebraskan(?) deposits or (?) Greenhorn Limestone |
10-31-1949 | 56 | 2,710 | 42 | 20 | 624 | 54 | 11 | 364 | 1,460 | 33 | 1.0 | 1.6 | 2,140 | 1,780 | 1,480 |
| cHays municipal wells | d51-57 | Terrace deposits | 1-5-1953 | 17 | 2.0 | 116 | 18 | 39 | 203 | 230 | 28 | .8 | 1.2 | 506 | 364 | 198 | ||
| c15-18-28da1 | 57.0 | Terrace deposits | 4-18-1949 | 23 | .25 | 157 | 25 | 43 | 235 | 340 | 26 | .8 | 3.9 | i88 | 494 | 301 | ||
| c15-18-28da2 | 53.0 | Terrace deposits | 4-5-1949 | 23 | 2.8 | 155 | 26 | 40 | 226 | 347 | 22 | .8 | 1.5 | 783 | 494 | 309 | ||
| 15-18-29dd1 | 18.8 | Terrace deposits | 10-31-1949 | 59 | 886 | 37 | .04 | 139 | 21 | 30 | 196 | 296 | 19 | .6 | 2.8 | 654 | 434 | 273 |
| c15-18-29dd2 | 52 | Terrace deposits | 9-1947 | 24 | 1.8 | 136 | 22 | 35 | 243 | 271 | 14 | .8 | 4.4 | 705 | 430 | 231 | ||
| 15-18-30ab | 13.6 | Crete Formation | 10-29-1949 | 61 | 1,430 | 41 | .20 | 206 | 30 | 78 | 336 | 296 | 42 | .4 | 204 | 1,060 | 638 | 362 |
| 15-18-33ba | 153.8 | Dakota Formation | 3-21-1947 | 63 | 3,590 | .00b | 18 | 13 | 770 | 334 | 264 | 860 | 3.6 | 2.0 | 2,100 | 98 | 0 | |
| 15-19-2aa | 17.3 | Nebraskan(?) deposits |
10-21-1949 | 60 | 1,320 | 37 | .50 | 117 | 32 | 110 | 304 | 164 | 97 | .5 | 130 | 904 | 424 | 175 |
| 15-19-4ad | 290.2 | Dakota Formation | 10-28-1949 | 58 | 1,430 | 23 | 4.7 | 77 | 25 | 208 | 304 | 306 | 125 | 1.4 | .6 | 908 | 295 | 46 |
| 15-19-7ab | 30.3 | Crete Formation | 10-22-1949 | 58 | 498 | 47 | .17 | 69 | 9.0 | 24 | 212 | 29 | 15 | .3 | 45 | 338 | 209 | 35 |
| 15-19-10ad1 | 28.6 | Alluvium | 10-29-1949 | 57 | 617 | 30 | .04 | 101 | 13 | 8.3 | 296 | 29 | 16 | .1 | 44 | 384 | 313 | 70 |
| 15-19-12aa | 16.2 | Nebraskan(?) deposits |
10-21-1949 | 59 | 785 | 40 | .10 | 86 | 22 | 61 | 355 | 80 | 21 | .4 | 41 | 520 | 305 | 13 |
| 15-19-27da | 156.3 | Dakota Formation | 3-21-1947 | 62 | 600 | .00b | 85 | 15 | 20 | 342 | 32 | 12 | .4 | 4.0 | 388 | 274 | 0 | |
| 15-19-28dd | 26.5 | Pleistocene undifferentiated |
10-22-1949 | 57 | 1,350 | 36 | .15 | 148 | 26 | 125 | 260 | 346 | 95 | .4 | 51 | 968 | 477 | 264 |
| 15-19-32cb | 32.3 | Pleistocene undifferentiated |
10-22-1949 | 58 | 1,210 | 43 | .16 | 163 | 30 | 80 | 316 | 366 | 32 | .7 | 20 | 880 | 530 | 271 |
| 15-20-7aa | 18.7 | Crete Formation | 10-29-1947 | 61 | 1,060 | 32 | .64 | 156 | 25 | 46 | 294 | 250 | 38 | .3 | 46 | 732 | 402 | 251 |
| 15-20-12ad | 32.5 | Crete Formation | 10-21-1949 | 58 | 913 | 29 | 3.1 | 130 | 14 | 18 | 244 | 8.0 | 45 | .1 | 184 | 628 | 382 | 182 |
| 15-21-5ab | 11.1 | Crete Formation | 10-29-1949 | 63 | 1,050 | 36 | .14 | 137 | 14 | 53 | 196 | 98 | 60 | .1 | 208 | 780 | 400 | 239 |
| 16-18-1Ocb | 168.2 | Dakota Formation | 10-29-1949 | 59 | 3,830 | 18 | 3.2 | 21 | 12 | 792 | 344 | 268 | 886 | 5.0 | .8 | 2,170 | 102 | 0 |
| a. One part per million is equivalent to 1 pound of substance per million pounds of water or 8.34 pounds per million gallons of water. b. In solution at time of analysis. c. Analysis by laboratory of Kansas State Board of Health. d. Composite from five wells (15-18-27ca, -27cb1, -27cb2, -28da1, -28da2). |
||||||||||||||||||
Table 5--Factors for converting parts per million to equivalents per million.
| Cation | Conversion factor | Anion | Conversion factor | |
|---|---|---|---|---|
| Ca++ | 0.0499 | HCO3- | 0.0164 | |
| Mg++ | .0822 | SO4-- | .0208 | |
| Na+ | .0435 | Cl- | .0282 | |
| K+ | .0256 | NO3- | .0161 | |
| F- | .0526 |
Chemical Quality of the Ground Water
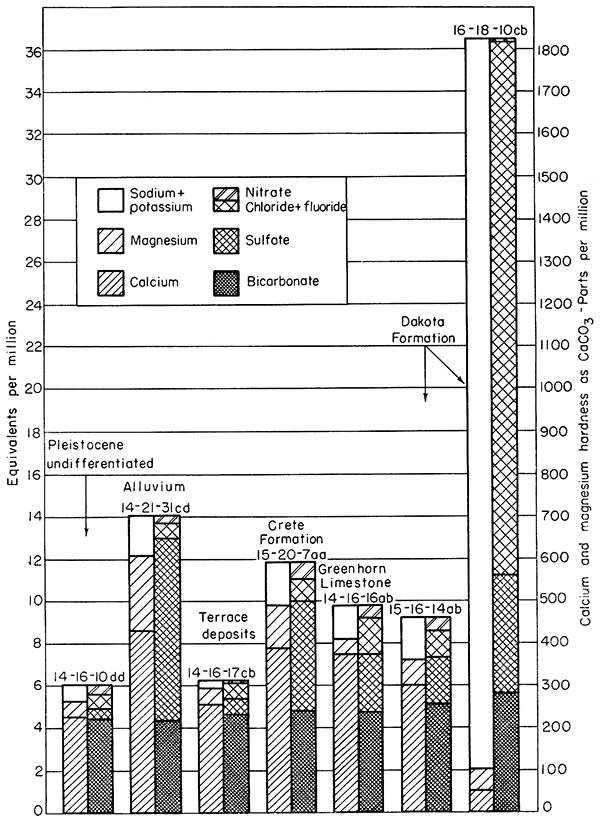
The chemical quality of ground water in the report area is indicated by analyses of water from 49 representative farm wells and from 7 municipal wells (Table 4). Factors for converting analytical results reported in parts per million to equivalents per million are given in Table 5. The relations of the principal mineral constituents in the water are shown graphically in Figure 16. Water samples from the farm wells were analyzed in the laboratory of the U. S. Geological Survey Quality of Water Branch in Lincoln, Nebraska; the water samples from the municipal wells and systems were analyzed by Howard Stoltenberg in the Laboratory of the Kansas State Board of Health. W. H. Durum, chemist, Quality of Water Branch, has prepared a preliminary report on the chemical quality of ground water in the area (1951).
Figure 16--Graphic representation of chemical analyses of water from wells in principal water-bearing formations.
Chemical Constituents in Relation to Use
The following discussion of the chemical constituents of ground water has been adapted from publications of the U. S. Geological Survey and the State Geological Survey of Kansas.
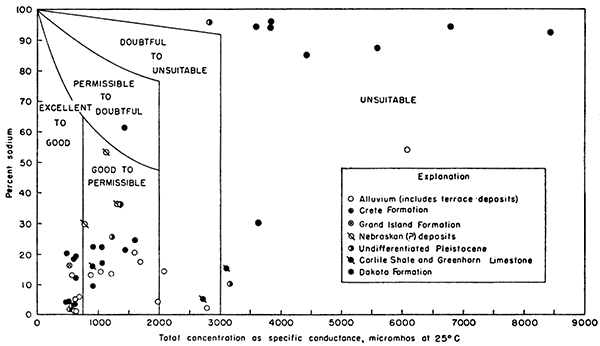
Dissolved solids--The residue left after a natural water has evaporated generally consists of mineral matter, some organic materials, and a small amount of water of crystallization. Except for difficulties resulting from hardness, water containing less than 500 ppm of dissolved solids generally is satisfactory for domestic use. Water having more than 1,000 ppm of dissolved solids is likely to contain enough of certain constituents to produce a noticeable taste Or to make the water unsuitable in other respects. Figure 17, adapted from Wilcox (1948), indicates that water having dissolved solids equivalent to a specific conductance of 3,000 micromhos or more (about 1,800 ppm for bicarbonate or chloride waters) is unsuitable for irrigation.
Figure 17--Classification of water for irrigation.
Dissolved solids in samples of water from wells ranged from 298 to 4,950 ppm. Samples from 16 wells contained less than 500 ppm, 18 samples contained between 500 and 1,000 ppm, 15 samples contained between 1,000 and 3,000 ppm, and 5 samples contained more than 3,000 ppm.
Hardness--The hardness of water is generally recognized by the amount of soap needed to produce a lather or suds and by the curdlike scum that forms during the washing process. Calcium and magnesium cause nearly all the hardness of water, and the carbonate and sulphate salts of these constituents form most of the scale in steam boilers and other vessels in which water is heated or evaporated.
The total hardness and the noncarbonate hardness of waters analyzed are given in Table 4, The noncarbonate hardness of water is caused by the presence of sulfates and chlorides of calcium and magnesium and is sometimes called "permanent" hardness because it can not be removed by boiling the water. "Temporary" hardness, caused by the presence of carbonates of calcium and magnesium in the water, can be removed by boiling the water.
Water having a hardness of less than 50 ppm is generally regarded as soft, and treatment for removal of hardness ordinarily is not necessary. Hardness between 50 and 150 ppm does not seriously interfere with the use of water for most purposes, but it increases the consumption of soap, and softening is profitable for laundries or other industries using large quantities of soap. Water having a hardness in the upper part of this range will cause considerable scale in steam boilers. Hardness exceeding 150 ppm is noticeable, and if the hardness is 200 or 300 ppm, the water for household use commonly is softened or cisterns are installed to collect rain water, When municipal water supplies are softened, hardness is generally reduced to about 100 ppm. Additional softening of a public supply is often judged not worth the increase in cost.
Samples of water from the report area ranged in total hardness from 53 to 1,840 ppm, and the noncarbonate hardness ranged from o to 1,540 ppm. Water samples from 4 wells had less than 150 ppm total hardness, 15 samples had between 150 and 300 ppm, 20 samples had between 300 and 500 ppm, 7 samples had between 500 and 1,000 ppm, and 8 samples had more than 1,000 ppm; 7 water samples had no noncarbonate hardness, 18 samples had between 1 and 100 ppm, 18 samples had between 100 and 500 ppm, 5 samples had between 500 and 1,000 ppm, and 6 samples had more than 1,000 ppm.
Iron--If iron is present in ground water in concentrations in excess of about 0.3 ppm, the excess will precipitate upon exposure to air. Water containing excess iron will have a noticeable discoloration and a disagreeable taste. Iron may be removed from most water by aeration and filtration.
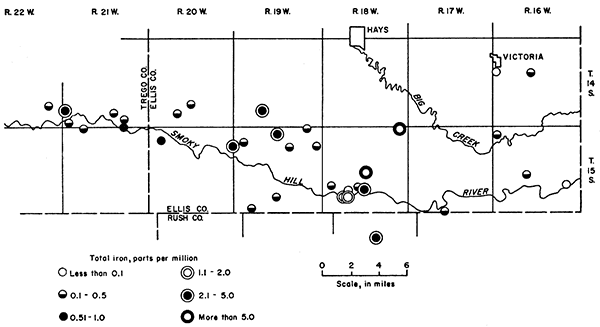
The concentrations of iron in 34 samples of water that are reported in Table 4 ranged from 0.03 to 20 ppm. Water samples from 18 wells contained less than 0.3 ppm, 6 samples contained between 0.3 and 1.0 ppm, 9 samples contained between 1.0 and 10.0 ppm, and 1 sample contained 20 ppm. The areal distribution of iron in ground water in the area is shown in Figure 18.
Figure 18--Map showing locations and limits of iron concentrations of selected water samples.
Chloride--Chloride, an abundant constituent of sea water, is normally present in small to moderate amounts in water from wells. Chloride is dissolved from rock materials, derived from sewage and industrial wastes, or results from sea water entrapped in marine rocks. Locally in this area, some chloride is derived from brine that is pumped from oil wells and is allowed to contaminate shallow ground-water supplies. Small concentrations of chloride have little effect on the suitability of water for most uses, but water containing more than about 500 ppm may have a salty taste. Water containing much chloride is unsuitable for irrigation and generally is corrosive to pipes and steam boilers.
Concentrations of chloride in water samples collected for chemical analysis ranged from 6.5 to 2,460 ppm; 45 samples contained less than 500 ppm, 4 samples contained between 500 and 1,000 ppm, and 5 samples contained more than 1,000 ppm. The water in well 15-18-20ab has been contaminated by oil-field brine and the sample contained 1,870 ppm of chloride.
Fluoride--Fluoride in concentrations of about 1.0 ppm helps prevent tooth decay, but fluoride in concentrations exceeding about 1.5 ppm may cause a discoloration known as "mottled enamel", which appears on the teeth of children who drink the water during the period when permanent teeth are forming. Fluoride in the water samples from wells in this area ranged from 0.1 to 7.0 ppm; 7 samples contained more than 1.5 ppm.
Nitrate--Recent discoveries (Comly, 1945; Metzler and Stoltenberg, 1950) indicate that excessive amounts of nitrate in water used in the preparation of food for infants may cause infant cyanosis.
The Kansas State Board of Health judges that waters containing more than 45 ppm of nitrate as NO3 are unsafe for use in infant feeding and that infant cyanosis is likely if the concentration of nitrate exceeds 90 ppm. Concentrations of nitrate in water samples from wells in the report area ranged from 0.6 to 348 ppm. Water samples from 12 wells contained more than 45 ppm nitrate and samples from 5 wells (14-21-34ba, 15-18-30ab, 15-19-2aa, 15-20-12ad, and 15-21-5ab) contained more than 90 ppm.
Some nitrate in ground water may be derived by solution of nitrate-bearing rocks, from the application of nitrate fertilizers, or from the growing of legumes in recharge areas, but strong concentrations may be due to seepage of surface water from a barnyard or other source of contamination. Table 6 is a comparison of the nitrate in water from 27 dug wells and 26 drilled or bored wells in the report area.
Table 6--Comparison of nitrate concentrations in water from dug and drilled wells.
| Range in concentration of nitrate (ppm) |
Dug wells | Drilled or bored wells | ||
|---|---|---|---|---|
| No. | Percent | No. | Percent | |
| 0-10 | 7 | 26 | 19 | 73 |
| 11-45 | 10 | 37 | 5 | 19 |
| 46-90 | 7 | 26 | 0 | 0 |
| More than 90 | 3 | 11 | 2 | 8 |
Sodium--For irrigation water the sodium content generally is expressed as the ratio of the concentration of sodium, in equivalents per million, to the total concentration of the cations (calcium, magnesium, sodium, potassium), This ratio, expressed as a percentage, is called "percent sodium". Water that has a large percent sodium, if used to irrigate certain types of soils, may deflocculate clays in the soil and cause the soil to become compacted. The percent sodium permissible in water for irrigation varies with the specific conductance, becoming smaller as the specific conductance increases (Fig. 17).
In the report area, water samples from wells ranged in percent sodium from 1 to 96. The percent sodium in 9 samples, including 8 from the Dakota Formation, was more than 60, but most of the water samples, except those from the Dakota Formation, are classed "excellent to good" or "good to permissible" for irrigation (Fig. 17).
Boron--Water containing less than 1.0 ppm boron generally is considered satisfactory for irrigating even such boron-sensitive crops as citrus trees, melons, and nut trees. None of the water samples analyzed contained more than 1.0 ppm.
Chemical Quality of Water in Relation to Geologic Source
The chemical quality of the ground water in the water-bearing formations has a wide range. In some formations it also varies widely in quality from place to place. In general, water from the different Pleistocene formations is similar in type and in degree of mineralization but differs from the water in the Dakota Formation, which is principally of the sodium chloride type, The quality of water in the principal water-bearing formations, the Dakota Formation, undifferentiated Pleistocene deposits, Crete Formation, terrace deposits, and alluvium, is discussed below and is illustrated graphically by Figure 16. The number of water samples collected from the Carlile Shale, Greenhorn Limestone, Grand Island Formation, and Nebraskan(?) deposits was not sufficient to indicate the quality of water in these formations.
Dakota Formation--Water from the Dakota Formation in this area generally is of the sodium chloride type, contains a large amount of dissolved solids, and is hard. A summary of several of the principal chemical constituents is given in Table 7. Total dissolved solids of 11 samples analyzed ranged from 388 to 4,950 ppm and averaged about 2,400 ppm. Except for well 15-18-9bb, which contained calcium sulfate type water, the total hardness ranged from 82 to 361 ppm and averaged 246 ppm, the lowest for the principal water-bearing formations. Six samples contained more than 1.5 ppm of fluoride, the greatest amount being 6.0 ppm from well 14-19-29dd, The fluoride concentrations of the 11 samples averaged 2.5 ppm.
Table 7--Summary of concentrations of some chemical constituents in 11 water samples from wells in Dakota Formation.
| Range in concentration (ppm) |
Number of samples | Range in concentration (ppm) |
Number of samples | |||
|---|---|---|---|---|---|---|
| Dissolved solids |
Chloride | Total hardness |
Nitrate | Fluoride | ||
| 0-200 | 0 | 3 | 3 | 0.0-1.5 | 2 | 5 |
| 201-500 | 1 | 1 | 7 | 1.6-5.0 | 6 | 5 |
| 501-1,000 | 2 | 3 | 0 | 5.1-10 | 1 | 1 |
| 1,001-2,000 | 0 | 3 | 1 | 10.1-20 | 1 | 0 |
| 2,001-3,000 | 4 | 1 | 0 | 21-40 | 1 | 0 |
| More than 3,000 | 4 | 0 | 0 | More than 40 | 0 | 0 |
Concentrations of chloride are considerably greater in samples from the Dakota Formation than in samples from the other aquifers, The concentrations ranged from 12 ppm for water from well 15-19-27da to 2,460 ppm for water from well 15-16-9aa. Only 2 water samples contained less than 100 ppm chloride, and 4 samples contained more than 1,000 ppm. The average of 11 samples was 880 ppm.
The concentrations of nitrate in the samples from the Dakota Formation are less than those from the other principal aquifers in the area, probably because most of the wells are drilled wells of small diameter, which are not as likely to be polluted by surface wastes as are larger diameter dug wells. Nine samples contained less than 10 ppm nitrate, and the greatest amount was 40 ppm.
Undifferentiated Pleistocene deposits--Undifferentiated Pleistocene deposits yield water to wells in two parts of the area shown on Plate 1, in a small area in the southwestern part of T. 15 S., R. 19 W., near the Smoky Hill School and in a more extensive "high terrace" area along Big Creek east and southeast of Hays. Analyses of 8 water samples from these deposits were made, 2 from wells in the Smoky Hill School area, and 6 from wells in the Big Creek area. Water from well 14-17-25aa was of the calcium chloride type and had a dissolved solids concentration of 1,690 ppm and a hardness of 1,390 ppm. Water from well 14-18-12bb was of the sodium chloride type and had 1,590 ppm dissolved solids, 7 ppm fluoride, and 53 ppm hardness, the lowest hardness of the samples analyzed. Durum (1951, p. 25) suggested that the quality of this water might have been affected by intrusion of altered brine from one of the nearby oil fields. The other 6 samples were calcium bicarbonate type similar to the water from the other Pleistocene deposits in the area. For these 6 samples, dissolved solids ranged from 298 to 968 ppm and averaged 531 ppm, total hardness ranged from 234 to 530 ppm and averaged 343 ppm, and the fluoride content was low. Water from well 15-19-28dd contained 51 ppm nitrate; the nitrate concentrations of the other samples were less than 31 ppm.
Crete Formation--Water samples from 11 wells tapping the Crete Formation in the "high terrace" area along Smoky Hill River were collected and analyzed. Water from wells 14-21-35ca, 15-18-30ab, and 15-29-7aa was of the calcium sulfate type and contained much bicarbonate. The sulfate is probably due to the solution of gypsum in the Carlile Shale in the vicinity of these wells. The other samples from the Crete Formation were of the calcium bicarbonate type and contained considerable hardness and dissolved solids but only a moderate amount of chloride and small amounts of fluoride and iron. Five samples contained more than 45 ppm of nitrate, and 2 samples contained more than 90 ppm. The 5 samples containing more than 45 ppm were from shallow wells ranging in depth from 12 to 33 feet, and the nitrate concentrations probably were caused by seepage from barnyards into the shallow aquifer. Table 8 is a summary of several of the principal chemical constituents in water from the Crete Formation.
Table 8--Summary of concentrations of some chemical constituents in 11 water samples from wells in Crete Formation.
| Range in concentration (ppm) |
Number of samples |
Range in concentration (ppm) |
Number of samples |
Range in concentration (ppm) |
Number of samples |
|||
|---|---|---|---|---|---|---|---|---|
| Dissolved solids |
Total hardness |
Chloride | Nitrate | Fluoride | Iron * | |||
| 0.0-5.0 | 0 | 1 | 0.0-0.1 | 4 | 1 | |||
| 300 or less | 0 | 5 | 5.1-20 | 3 | 1 | 0.11-0.5 | 7 | 5 |
| 301-500 | 6 | 4 | 21-45 | 6 | 4 | 0.51-1.5 | 0 | 1 |
| 501-1,000 | 3 | 2 | 46-90 | 2 | 2 | 1. 51-10 | 0 | 1 |
| More than 1,000 | 2 | 0 | More than 90 | 0 | 3 | More than 10 | 0 | 0 |
| * Total iron determined for only 8 samples. | ||||||||
Alluvium and terrace deposits--Sixteen water samples from wells in the alluvium and associated late Pleistocene terrace deposits were collected for analysis. Four water samples were of the bicarbonate type; one sample of water (from well 15-18-20ab) contaminated with oil-field brine was of the chloride type; and 11 samples were of the sulfate type. Concentration of sulfate in water in the alluvium is most common in the western part of the area and in the Hays well-field area near Schoenchen. As in the Crete deposits, the sulfate is probably derived from gypsum in the adjacent Carlile Shale.
Water in the alluvium and terrace deposits is hard and relatively strongly mineralized. Except for well 15-18-20ab, the total hardness ranged from 295 to 1,130 ppm and averaged 564 ppm; dissolved solids ranged from 383 to 1,660 ppm and averaged 827 ppm. The fluoride and chloride contents were low, and the percent sodium was generally less than 20. The concentration of iron was excessive, only 3 samples out of 14 had less than 0.1 ppm iron, and 4 samples had more than 1.0 ppm. Water from wells 14-20-28ad (46 ppm nitrate), 14-21-30ca (55 ppm) and 14-21-34ba (348 ppm) contained more than 45 ppm nitrate. The nitrate concentrations in water from these wells may be the result of infiltration of water to the ground-water reservoir or well from barnyards or other sources of contamination.
A summary of the principal chemical constituents in water in the alluvium and terrace deposits is given in Table 9.
Table 9--Summary of concentrations of some chemical constituents in 16 water samples from wells in alluvium and terrace deposits.
| Range in concentration (ppm) |
Number of samples |
Range in concentration (ppm) |
Number of samples |
Range in concentration (ppm) |
Number of samples |
||
|---|---|---|---|---|---|---|---|
| Sulfate | Dissolved solids |
Total hardness |
Nitrate | Iron * | |||
| 101-200 | 0 | 0 | 0 | 11-20 | 2 | 0.1-1.0 | 6 |
| 201-300 | 4 | 0 | 1 | 21-45 | 3 | 1.1-5.0 | 4 |
| 301-500 | 4 | 4 | 8 | 46-90 | 2 | 5.1-10.0 | 0 |
| 501-1,000 | 3 | 7 | 4 | 91-200 | 0 | 10.1-20 | 0 |
| 1,001-2,000 | 1 | 4 | 3 | More than 200 | 1 | More than 20 | 0 |
| More than 2,000 | 0 | 1 | 0 | ||||
| * Total iron determined for only 13 samples. | |||||||
Pollution by Oil-Field Brines
Considerable brine is produced with crude petroleum pumped from the oil fields of the area. Some oil wells produce only a small amount, whereas others, especially old wells from which a large part of the recoverable oil has been pumped, may produce hundreds of barrels of brine daily. During the early period of oil field development, most of the brine was discharged into shallow earth "evaporation ponds". The principle of the evaporation pond was that the brine was removed from the pond by evaporation, which is relatively rapid in this area during the summer. Just how the mineral matter in the brine was to be removed is not clear, inasmuch as evaporation removes only the water and leaves the mineral matter behind in the increasingly concentrated brine. Frye and Brazil (1943) indicated that evaporation ponds are considered safe for the disposal of salt water only in areas where the material forming the sides and floor of the pond is impervious rock, such as shale. Ponds are unsatisfactory for the disposal of brine where the near-surface materials are pervious and contain fresh water, as do the alluvium or Pleistocene terrace deposits, because in such places the salt water percolates downward and laterally into the porous material and contaminates the fresh-water supply.
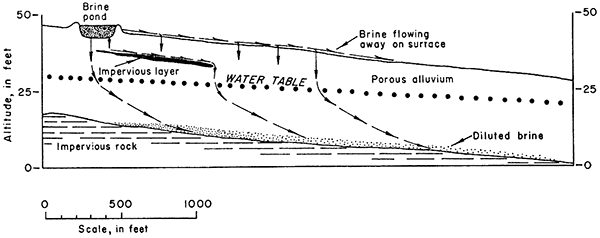
In the oil fields of this area, evaporation ponds have been constructed of shale and other impervious material, but in alluvial valleys many of the ponds are constructed of permeable alluvium. Brine may seep out of these ponds and contaminate the fresh water in the alluvium or flow away on the surface to contaminate the surface streams in the area. Figure 19 indicates the theoretical movement of brine from a pond of porous material into the ground-water body in the alluvium and into the surface drainage.
Figure 19--Diagrammatic section showing theoretical movement of oil-field brine.
Contamination in Ruder Creek valley--Brine produced with oil in the Ruder oil pool in the central part of T. 15 S., R. 18 W. (Fig. 4), began to contaminate ground water in the alluvium of the valley soon after the field was put into production in 1936. By 1951, the alluvium for 2 miles below the oil pool was contaminated with brine, and contaminated ground water seemed to be moving down the valley toward Smoky Hill River. The water sample from well 15-18-20ab is an example of water that has been contaminated by oilfield brine. The contamination in this tributary valley was the result of the method formerly used for disposing of brine in shallow ponds from which it seeped into the alluvium, overflowed into surface streams, or seeped through the walls of the pond and flowed away on the land surface. Since 1951 brine pumped with the oil in the Ruder pool has been pumped into deep wells, and the contamination of the water in the alluvium gradually is decreasing as fresh water from precipitation flushes out the brine, Many years must elapse, however, before the effects of contamination disappear.
Possible contamination in other oil-field areas--As previously quoted by Frye and Brazil, disposal of oil-field brines in surface ponds is safe only where the ponds are constructed of impervious materials. In the report area impervious clays are formed by weathering of the Carlile Shale and by soil formation in the late Pleistocene silts, Brine ponds constructed of these materials would probably not allow subsurface seepage or outflow through the pond walls and would be satisfactory in this respect. The mineral constituents that cause the contamination of water, however, will not be evaporated from any pond; therefore, such ponds are potential sources of ground-water contamination because of the possibility that they might overflow during periods of heavy precipitation and carry minerals into ground water during recharge. If they are abandoned, the concentration of mineral salts in the ponds will be left as potential sources of contamination.
Ground Water Regions and Areas
Ground-water regions and areas are shown on Figure 20, and the general availability of water in each region or area is given in Table 10. The report area is divided into three ground-water regions: the uplands, the Smoky Hill and Big Creek terraces, and the main valleys. Regions in which ground-water conditions are not uniform are divided into ground-water areas.
Figure 20--Ground-water regions and areas.
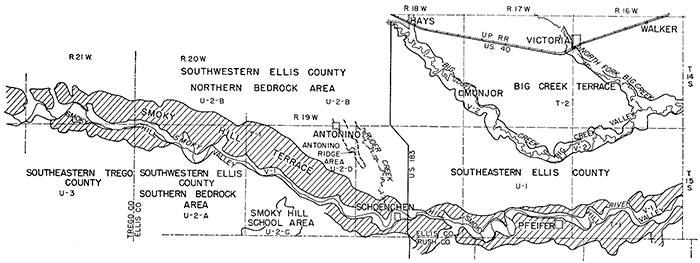
Table 10--General availability of ground water by areas.
| Region and area | General availability of water |
Principal geologic source |
Depth to water, feet |
General quality of ground water |
Remarks | ||
|---|---|---|---|---|---|---|---|
| (U) Upland regions: | |||||||
| (U-1) Southeastern Ellis County. | Small supplies. | Dakota Formation, alluvium. |
Dakota: average 112. Alluvium: 18. |
Dakota: moderately high in Na, Cl, Fl, Fe. Alluvium: hard. |
Dakota: artesian conditions; average well depth, 190 feet. | ||
| (U-2) Southwestern Ellis County upland bedrock areas. |
|||||||
| (U-2-A) Southern bedrock area. | Small to moderate supplies. | Dakota Formation, alluvium. |
Dakota: 300 or more. Alluvium: 30 or less. |
No data. | Reported data only. | ||
| (U-2-B) Northern bedrock area. | Moderate supplies. | Dakota Formation, alluvium. |
Dakota: average about 160. Alluvium: 30 or less. |
Dakota: soft. high in Fl, Fe. Alluvium: hard. |
|||
| (U-2-C) Smoky Hill School area. | Moderate supplies. | Undifferentiated Pleistocene. |
20 to 30. | No data. | Water-table conditions. | ||
| (U-2-D) Antonino ridge area. | Moderate supplies. | Nebraskan(?) deposits. |
15 or less. | Hard. | Alluvial deposits capping small ridge. Water-table conditions. |
||
| (U-3) Southeastern Trego County. | Small supplies locally. |
Dakota Formation, alluvium. |
Dakota: 300 or more. Alluvium: 20 or less. |
No data. | Many areas without water supplies. | ||
| (T) High terrace regions: | |||||||
| (T-I) Smoky Hill terrace. | Moderate supplies, except locally. |
Crete Formation, alluvium. |
10-50, average about 30. |
Hard, high in Fe. | Shale bedrock locally above water table. | ||
| (T-2) Big Creek terrace. | Moderate supplies. | Undifferentiated Pleistocene deposits. |
12-55, average about 30. |
Hard, otherwise good. | Water-table conditions. | ||
| (V) Main valley areas: | |||||||
| (V-I) Smoky Hill Valley. | Small to large supplies. |
Terrace deposits and alluvium. |
Generally less than 20. |
Hard. | Yielded more than 700 gpm in tested wells. |
||
| (V-2) Big Creek Valley. | Small supplies. | Terrace deposits and alluvium. |
20 to 30. | Hard. | |||
Upland Regions (U)
Three ground-water regions are of the upland type: the southeastern Ellis County upland, the southwestern Ellis County upland, and southeastern Trego County upland. In general, upland regions are areas of shale bedrock locally covered by thin layers of loess or colluvium. Water supplies are not abundant and generally are restricted to the alluvium of small valleys or to deep wells drilled into the Dakota Formation.
Southeastern Ellis County upland (U-1)--In the southeastern Ellis County upland the Cretaceous bedrock lies at or near the surface. The region is characterized by gently rolling hills, by relatively flat minor divides, and by steep bluffs where it borders Smoky Hill River and Big Creek valleys. The region is poorly supplied with ground water and in many places has no potable ground-water supplies, Locally, wells drilled nearly 200 feet to the sandstones of the Dakota Formation yield small to moderate supplies of mineralized water, In many places, water in the Dakota Formation is too mineralized to be suitable for human or livestock use. Shallow wells of small yield have been constructed in the alluvium along some of the small streams, but these wells go dry during prolonged periods of drought, hence are not dependable sources of ground water. At a few places where the Greenhorn Limestone and Carlile Shale are deeply weathered, shallow wells obtain a small yield from these formations. In general, water supplies in the southeastern Ellis County upland are small, undependable, or strongly mineralized.
Southwestern Ellis County upland bedrock areas (U-2)--The southwestern Ellis County upland is the region that includes the strongly dissected upland south of Smoky Hill River in the southwestern corner of the county (U -2-A), and the shale slopes, pediment slopes, and gently rolling upland adjacent to the prominent terrace north of Smoky Hill River (U-2-B), Except for two small areas (C and D) discussed below, the region is not well supplied with ground water. Dependable wells of moderate yield may be drilled in the alluvium of the larger tributary streams, but in the alluvium of the smaller tributaries only small supplies can be obtained. Locally, wells 300 to 500 feet deep drilled to the Dakota Formation yield moderate supplies of ground water suitable for domestic and stock use, Elsewhere, areas of several square miles, such as the east-central part of T, 15 S., R. 20 W., are devoid of any potable ground-water supplies because the Dakota Formation contains strongly mineralized water and no alluvial deposits are present.
Smoky Hill School area (U-2-C)--Several farms and one rural school in an area of about 4 square miles in the southwestern part of T, 15 S., R. 19 W., Ellis County, and several square miles in northern Rush County are supplied from a body of ground water in undifferentiated Pleistocene alluvial deposits, The depth to water is 20 to 30 feet. Ground water moves northeastward at a hydraulic gradient of about 20 feet per mile. Yields are moderate and supplies are dependable.
Antonino ridge area (U-2-D)--Ground water is found at depths generally less than 15 feet in Pleistocene (Nebraskan?) alluvial deposits under the narrow ridge extending southeastward through Antonino to the center of sec. 18, T. 15 S., R. 18 W. Wells of small to moderate yields supply the residents of Antonino and several farms to the southeast, No large yields are reported, but the supply is dependable,
Southeastern Trego County (U-3)--Except for the valley area and the Smoky Hill terrace region, discussed below, southeastern Trego County is an upland region of flat-topped, steep-sided, strongly dissected hills underlain by the Niobrara Chalk. This region is almost devoid of ground-water supplies, except for a few wells that tap the Dakota Formation, reported to be more than 500 feet deep, and for wells of small yield tapping alluvial deposits in small tributary valleys.
High Terrace Regions (T)
Smoky Hill terrace (T-1)--The Smoky Hill terrace region is the flat to gently rolling, slightly dissected region adjacent to Smoky Hill River valley. It is underlain by the Crete Formation and locally by the Grand Island Formation and in many places is separated from the valley by shale banks. Except where the shale bedrock protrudes above the water table, moderate ground-water supplies are available from the Pleistocene deposits throughout the region, Ground water in the Crete Formation moves toward Smoky Hill River and downstream. The hydraulic gradient ranges from 20 to 40 feet per mile but averages about 35 (Pl. 2), Wells average about 30 feet deep, have small to moderate yields, and are dependable,
Big Creek terrace (T-2)--The Big Creek terrace lies principally north of Big Creek in east-central Ellis County; the surficial rocks are unconsolidated alluvial deposits. Moderate supplies of ground water under water-table conditions are found at shallow depths in the region. Depth of wells ranges from 12 to 55 feet and averages about 30 feet. The principal aquifers are stream-deposited sand and gravel of undifferentiated Pleistocene age. In general, ground water moves from the divides toward the principal streams, Big Creek and North Fork Big Creek. The hydraulic gradient ranges from about 12 to 50 feet per mile and averages about 25 feet per mile (Pl. 2). The ground water is hard but serves all domestic and farm uses.
Main Valley Areas (V)
The inner valley of Smoky Hill River, which is mapped as alluvium on Plate 1, and the valley of Big Creek are the two main valley areas.
Smoky Hill Valley (V-1)--The Smoky Hill Valley area, a flat area 1/4 to 1 mile wide bordering the stream, generally is well supplied with ground water under water-table conditions. Alluvium and Wisconsinan terrace deposits are as much as 65 feet thick, and as much as 40 feet is saturated. The material generally is coarse sand and gravel sufficiently permeable to yield water readily to wells, Yields of more than 700 gallons a minute were reported during test-pumping of wells near the Hays municipal well field. Depth to water is generally less than 20 feet. Where the thickness of saturated material is less, yields are proportionately smaller. Ground water moves downstream and toward the river and discharges into the stream. Generally the water contains iron and is hard but is used for domestic and farm purposes.
Big Creek Valley (V-2)--Big Creek Valley is the flat, terraced area adjacent to the creek. The width is 1/2 to 1 mile and averages about 3/4 mile. Ground water occurs under water-table conditions in alluvial deposits adjacent to the stream. Ground water moves into these deposits from the high terrace deposits, principally north of the valley, and within the alluvial deposits it moves generally downstream. The alluvial deposits are principally sand, gravel, and silt; the lower saturated material consists principally of gravel locally intermixed with silt. The saturated thickness and permeability are not great, and yields of wells generally are small. The depth to water averages about 20 feet.
Prev Page--Geomorphology || Next Page--Well Records
Kansas Geological Survey, Geology
Placed on web Feb. 4, 2013; originally published March 1961.
Comments to webadmin@kgs.ku.edu
The URL for this page is http://www.kgs.ku.edu/Publications/Bulletins/149/06_gw.html