Prev Page—Utilization of Water || Next Page—Geologic Formations
Ground Water, continued
Chemical Character of Water
The general chemical character of the ground waters in Scott County is indicated by the analyses of samples of water from 29 wells and one spring distributed as uniformly as practicable within the county and among the principal water-bearing formations (Table 15). The wells include the water supply at Scott City (well 112.) The samples of water were analyzed by Elza H. Holmes, chemist in the Water and Sewage Laboratory of the Kansas State Board of Health.
Chemical Constituents in Relation to Use
The following discussion of the chemical constituents of ground water in relation to use has been adapted from publication of the U.S. Geological Survey.
Dissolved Solids—When water is evaporated, the residue that is left consists mainly of the mineral constituents listed below and generally includes a small quantity of organic matter and a little water of crystallization. Waters containing less than 500 parts per million of dissolved solids generally are entirely satisfactory for domestic use except for difficulties rising from the hardness or occasionally excessive content of iron. Waters containing more than 1,000 parts per million are likely to include enough of certain constituents to produce a considerable taste or to make the water unsuitable in some other respect.
The dissolved solids in the samples of ground water collected in Scott County are given in Table 16. This table indicates most of the ground waters in Scott County contain total dissolved solids ranging from 200 to 400 parts per million. Five samples of water contained total dissolved solids of more than 400 parts per million.
Table 16—Summary of the chmical character of the samples of water from typical wells in Scott County.
| Range in parts per million |
Number of samples | |||
|---|---|---|---|---|
| Alluvium | Ogallala | Ogallala and/or Niobrara |
Pleistocene and/or Ogallala |
|
| Dissolved Solids | ||||
| 101 - 200 | 2 | |||
| 201 - 300 | 12 | 1 | 4 | |
| 301 - 400 | 5 | |||
| 401 - 500 | 1 | 1 | ||
| 501 - 600 | 1 | 2 | ||
| More than 600 | 1 (a) | |||
| Hardness | ||||
| 101 - 200 | 8 | 2 | ||
| 201 - 300 | 10 | 2 | 3 | |
| 301 - 400 | 1 | 1 | 1 | |
| 401 - 500 | 1 | |||
| More than 500 | 1 (b) | |||
| Fluoride | ||||
| Less than 0.5 | 2 | |||
| 0.6 - 1.0 | 1 | 2 | 2 | 2 |
| 1.1 - 1.5 | 1 | 1 | 1 | |
| 1.6 - 2.0 | 6 | 2 | ||
| 2.1 - 2.5 | 6 | 1 | ||
| 2.6 - 3.0 | 2 | 1 | ||
| Iron | ||||
| Less than 0.19 | 10 | 4 | ||
| 0.2 - 0.5 | 2 | 1 | 1 | |
| 0.6 - 1.0 | 2 | |||
| 1.1 - 1.5 | 2 | 1 | ||
| 1.6 - 2.0 | 1 | 1 | ||
| 2.1 - 5.0 | 1 | 2 | 1 | |
| 5.1 - 8.0 | 1 | |||
a. 2,202 parts per million
b. 1,339 parts per million
Hardness—The hardness of water is commonly recognized by the increase in the amount of soap needed to produce a lather and by the curdy precipitate that forms before a permanent lather is obtained. Calcium and magnesium are the constituents that cause practically all the hardness of ordinary waters and are also the active agents in the formation of the greater part of all the scale formed in steam boilers and in other vessels in which the water is heated or evaporated.
In addition to the total hardness, the table of analyses shows the carbonate hardness and the noncarbonate hardness. The carbonate hardness is that due to the presence of calcium and magnesium bicarbonates and can be almost entirely removed by boiling. In some reports this type of hardness is called temporary hardness. The noncarbonate hardness is due to calcium and magnesium sulfates or chlorides. It cannot be removed by boiling and has sometimes been called permanent hardness. With reference to use with soaps, there is no difference between carbonate and noncarbonate hardness. In general the noncarbonate hardness forms harder scale in steam boilers.
Water having a hardness of less than 50 parts per million is generally rated as soft, and its treatment for the removal of hardness is rarely justified. Hardness between 50 and 150 parts per million does not seriously interfere with the use of water for most purposes, but it does slightly increase the consumption of soap and its removal by a softening process is profitable for laundries or other industries that use large quantities of soap. Treatment for the prevention of scale is necessary for the successful operation of steam boilers that use water in the upper part of this range of hardness. Hardness of more than 150 parts per million can be noticed by anyone. Where public supplies are softened, an attempt is generally made to reduce the hardness to about or less than 80 parts per million. The additional improvement from further softening of a public supply is not deemed worth the additional cost. The hardness of the 30 samples of water that were analyzed is given in Table 16. This table indicates that most of the ground waters in Scott County range in hardness from 100 to 300 parts per million. Ten samples had hardness ranging from 100 to 200 parts per million and 115 samples had hardness ranging from 200 to 300 parts per million. The other five samples of water that were analyzed had a hardness of more than 300 parts per million.
Iron—Next to hardness, iron is the constituent of natural waters that in general receives the most attention. The quantity of iron in ground waters may differ greatly from place to place even in waters from the same formation. If a water contains much more than 0.1 part per million of iron, the excess may separate out after exposure to the air and settle as a reddish sediment. Iron which may be present in sufficient quantity to give a disagreeable taste and to stain cooking utensils may be removed from most waters by simple aeration and filtration but a few waters require the addition of lime or some other substance.
The iron content of the samples of ground water that were analyzed is given in Table 16. About half of the samples of water collected in Scott County contained less than 0.2 parts per million of iron. The other samples contained iron ranging up to more than 5 parts per million.
Fluoride—Although determinable quantities of fluoride are not so common as fairly large quantities of other constituents of natural waters, it is desirable to know the amount of fluoride present in waters that are likely to be used by children. Fluoride in water has been shown to be associated with a dental defect known as mottled enamel, which may appear on the teeth of children who drink water containing fluoride during the period of formation of the permanent teeth. It has been stated that waters containing more than 1 part per million of fluoride are likely to produce mottled enamel (Dean, 1936, pp. 1,269-1,272). If the water contains as much as 4 parts per million of fluoride, most of the children exposed are likely to have mottled enamel. Small quantities of fluoride, not sufficient to cause mottled enamel, are likely to be beneficial by decreasing-dental caries (Dean, Arnold, and Elvove, 1942, pp. 1,155-1,179).
The fluoride content of the samples of ground water that were analyzed is given above in Table 16. Of the 30 samples of water collected in Scott County, nine contained less than one part per million of fluoride, 11 contained between 1 and 2 parts per million of fluoride, and 10 samples contained more than 2 parts per million of fluoride.
Water for Irrigation—The suitability of water for use in irrigation is commonly believed to depend mainly on the total quantity of soluble salts and on the ratio of the quantity of sodium to the total quantity of sodium, calcium, and magnesium together. The quantity of chloride may be large enough to affect the use of the water and in some areas other constituents such as boron may be present in sufficient quantity to cause difficulty. In a discussion of the interpretation of analyses with reference to irrigation in Southern California, Scofield (1933) states that if the total concentration of the dissolved salts is less than 700 parts per million there is not much probability of harmful effects in irrigation use. If it exceeds 2,100 parts per million, there is a strong probability of damage to the crop or to the land or to both. Water containing less than 50 percent sodium (the percentage being calculated as 100 times the ratio of the sodium to the total bases, in equivalents) is not likely to be injurious but if it contains more than 60 percent its use is inadvisable. Similarly, a chloride content of less than 142 parts per million is not objectionable, but more than 355 parts per million is undesirable. It is recognized that the harmfulness of irrigation water is so dependent on the nature of the land and the crops and on the manner of use and the drainage that no hard and fast limits can be adopted.
All but one of the samples of water collected in Scott County are within the limits suggested by Scofield for safe waters for use in irrigation. The sample of water from well 70 contained 2,202 parts per million of total dissolved solids and it is probable that this water would not be suitable for irrigation. All of the samples of water collected from wells receiving their entire supply from the Ogallala formation are well within the limits suggested by Scofield.
Sanitary Considerations
The analyses of water given in Table 15 show only the amount of dissolved mineral matter in the water and do not indicate the sanitary quality of the water. An abnormal amount of certain mineral matter, such as nitrate, however, may indicate pollution of the water.
Dug wells and springs are more likely to become contaminated than are properly constructed drilled wells but great care should be taken to protect from pollution every well and spring used for domestic and public supply. It is important that the top of the casing be sealed in such a manner as to prevent surface water from entering the well and where pump pits are used, the top of the casing should extend above the floor of the pit so that surface water cannot drain into the well. In constructing wells equipped with ordinary lift or force pumps, it is a good plan to allow the casing to extend several inches above the platform so that the pump base will set down over the top of the casing thus effecting a tight seal. If the casing is left flush with the top of the platform, opportunity is afforded for drainage into the well and for possible contamination. Wells should not be located where there are possible sources of contamination such as drainage from the vicinity of buildings or cesspools.
Chemical Character in Relation to Water-bearing Formations
Nearly all the water wells in Scott County obtain water from the sand and gravel beds of the Ogallala formation but a few wells draw water from the alluvial deposits along the valleys and from the Niobrara formation in the northern and southern parts of the county. Some wells penetrate water-bearing material in both the alluvium and the Ogallala formation, the Niobrara and Ogallala formations, and the Pleistocene deposits and the Ogallala formation.
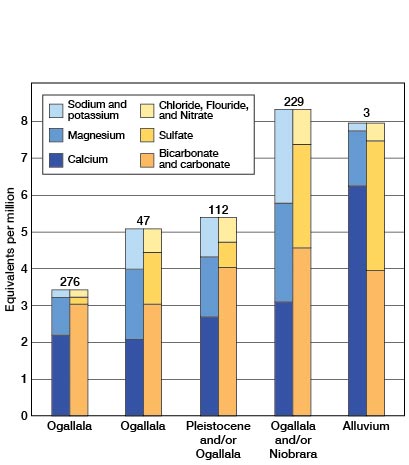
The quality of the water in relation to the water-bearing formations is summarized in Table 16 and shown graphically in Figure 16. The Pliocene deposits, comprising the Ogallala formation, and the sands and gravels of Pleistocene age yield water to most of the wells in this area. Samples of water from these deposits range in hardness from 162 to 344 parts per million; the average hardness is about 240 parts per million. The total dissolved solids in waters from the Pliocene and Pleistocene sediments ranged from 169 to 366 parts per million. The waters from the alluvium and the Niobrara formation are very hard and contain high amounts of dissolved solids. Fifteen of the 19 samples of water from wells in the Ogallala formation contained more than one part per million of fluoride indicating that this water may be harmful to children's teeth.
Figure 16—Analyses of waters from the principal water-bearing formations in Scott County.
Prev Page—Utilization of Water || Next Page—Geologic Formations
Kansas Geological Survey, Geology
Comments to webadmin@kgs.ku.edu
Web version March 2003. Original publication date July 1947.
URL=http://www.kgs.ku.edu/General/Geology/Scott/05_gw6.html