Kansas Geological Survey, Open-file Report 1999-44
by
Margaret Townsend
KGS Open-file Report 1999-44
High nitrate-nitrogen concentration has been measured in the ground water around the city of Pretty Prairie, Kansas. A previous study in the area surrounding Pretty Prairie showed that the primary source of nitrate was from fertilizer (Dealy, 1995). The previous study used complete chemical analyses, δ15N, and atrazine and its degradation products as indicators of nonpoint source contamination from agricultural sources.
The current study used complete analyses and δ15N to try to determine sources for the observed high nitrate-nitrogen concentrations in two city wells and in irrigation wells surrounding the city. Most of the land immediately to the north, west, and south of the city of Pretty Prairie is in irrigated agriculture. There is a golf course to the west of the city. A church well was sampled to the east of town. This well was the only domestic well sampled in this study. Two city wells were sampled, one at the northeast edge of town that was drilled in 1994, the other was in the center of town and was not used except occasionally because of the continued high nitrate-nitrogen values measured in the well.
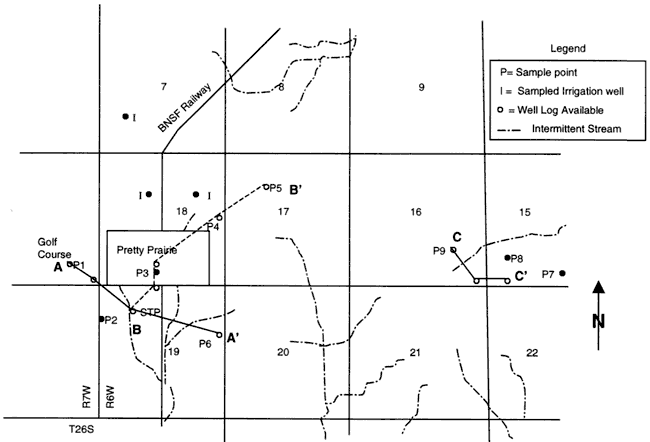
Figure 1 shows the location of the wells sampled for this study. The area of study is T. 26 S., R. 6-7 W., Sections 7, 15, 16, 17, 18, 20, 21 in Reno County, Kansas.
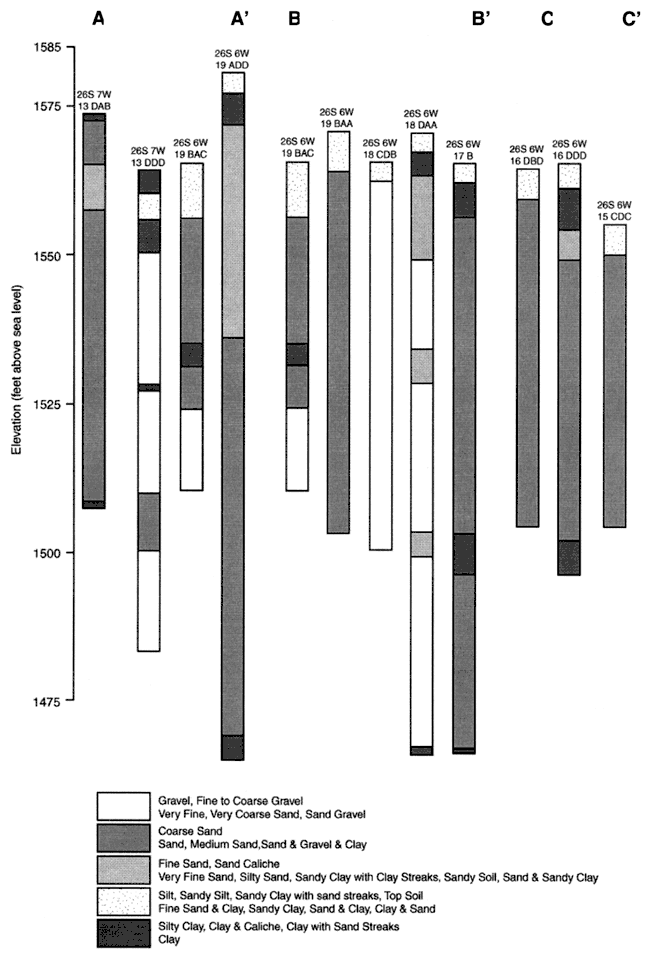
Figure 1. Location of wells sampled in August, 1999. Rectangle is city of Pretty Prairie, KS. Area is in T. 26 S., R. 6-7 W. in Reno County, Kansas. Cross-sections of well-logs shown by A-A', B-B', C-C' presented in Appendix B.

Sites for water sampling were generally recent irrigation or city wells and had water-well logs (WWC5 from the Kansas Department of Health and Environment (KDHE) available. Several sites were selected east of the city to determine if there was lower nitrate water present in that direction. The sites circle the city in all directions (Figure 1).
All samples were analyzed for major cations (calcium, magnesium, sodium, potassium) and anions (chloride, bicarbonate, sulfate, nitrate, and fluoride), in addition to pH, specific conductance, temperature, and calculated total dissolved solids by the Analytical Services section of the Kansas Geological Survey. All of the complete analyses were within acceptable error (appendix A, table A-1). A sample was also collected for nitrogen-15 (15N) isotope analysis. This analysis was performed at the University of Virginia Department of Environmental Sciences.
Water samples were collected from 6 irrigation, 2 public water supply, and one domestic well in August, 1999. Three additional irrigation wells in the area were sampled in July, 1999. Water samples were collected in clean polyethylene bottles of 500-ml, 250-ml, and 125-ml sizes. The 250-ml bottle was spiked with 2% HCL acid as a preservative for the nutrient samples. The bottle was marked at the 200-ml level and sample was collected to that mark.
Water samples were collected after specific conductance and temperature measurements had stabilized for at least 5 minutes. pH measurements also were measured until the same value was read to within 0.1 pH unit. Once the measurements had stabilized, the samples were collected in labeled bottles and placed in coolers where they were kept on ice until the return to KGS in Lawrence, KS. The samples were then stored in a refrigerator or in a freezer until time for analysis.
The samples for nitrogen-15 analysis were collected in the 125-ml bottles and kept on ice until the return to Lawrence. Then the samples were placed in a freezer until sent by Federal Express to Dr. Steve Macko at the University of Virginia. The samples were analyzed using a GC Mass Spec method developed by Dr. Macko. All analyses were completed within a month of collection.
The area is generally flat with topographic elevation changes from 1600 feet on the western edge of the study area to 1550 feet at the eastern edge. The geology of the area consists of Quaternary Pleistocene sand and gravel units. Evaluation of well logs in the local area indicates that there is minimal clay within the soil and stratigraphic profile of the aquifer. The predominant sediment is medium sand and coarse sand and gravel (Bayne, 1956). Depth of wells sampled in the area of study ranged from 51 to 103 feet. Sample well-logs for the area are shown in Appendix B. Lines of section shown in figure 1. Principle aquifer material is sand and gravel mixed together. The presence of highly transmissive materials suggests rapid movement of water from the land surface to ground water.
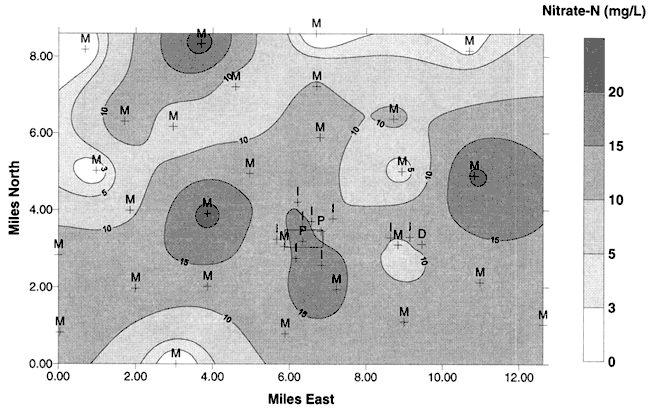
Average water-level elevations measured during the previous study in the area (Dealy, 1995) indicate a general flow direction to the north-northeast (Figure 2) . The data are somewhat sparse in the vicinity of Pretty Prairie as none of the irrigation or city wells were measured for water levels.
Figure 2. Ground-water flow directions and water-table elevations from average values measured in 1995 study (Dealy, 1995). Flow is generally to the northeast. The letter M indicates a monitoring well. Unlabeled crosses are sites sampled in this study. Dark rectangle is the city of Pretty Prairie, KS. Water-level measurements were not collected at the time of water sampling.

Surface drainage in the area consists of intermittent streams, which flow to the south (Figure 1). The low topographic relief in the area, plus the limited surface drainage, suggests that recharge to the aquifer by rainfall or irrigation is either lost due to evapotranspiration or plant use, or moves quickly downward through the unsaturated zone. The potential for runoff to streams is low. There is minimal clay in the unsaturated zone and aquifer to retard the downward movement of water, and any contaminants carried by the water, to the ground water.
The city of Pretty Prairie has records of nitrate-nitrogen values above the drinking water limit of 10 mg/L. The city has had several agencies collect samples and try to determine the source of the nitrate. The Equus Beds Groundwater Management District #2 did a study with Dr. Roy Spalding of the University of Nebraska using δ15N and atrazine plus atrazine degradation products in 1992-1994 to determine if the observed nitrate was from agricultural sources. The results of that study indicated that the nitrate was from fertilizer sources in a predominantly dryland farming area.
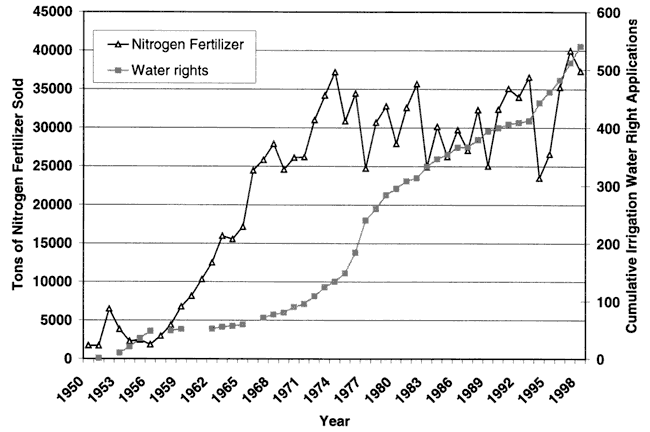
Figure 3 shows the cumulative irrigation water rights and tonnage of nitrogen fertilizer sold in Reno County. The figure shows an increasing trend in both fertilizer and potential water use in the county suggesting that irrigated farming has increased over time.
Figure 3. Graph of total tonnage of nitrogen fertilizer and cumulative irrigation water rights application for Reno County.
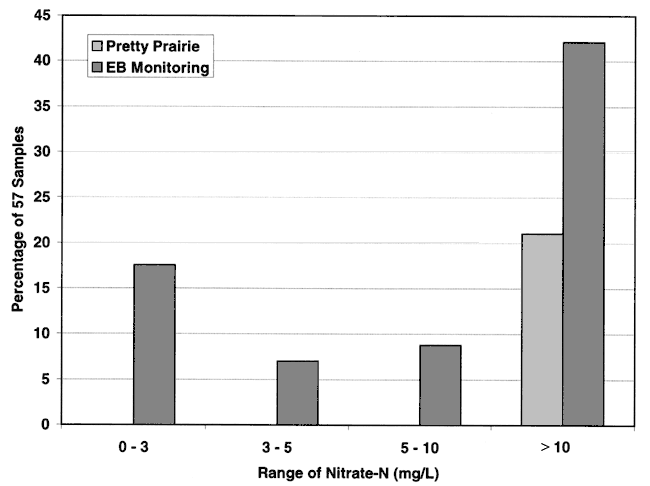
This study used irrigation wells, city wells, and one domestic well surrounding the city for water-quality sampling (Figure 1). Figure 4 shows the distribution of nitrate-nitrogen for the 12 samples collected in this study and the Equus Beds monitoring wells from the 1995 study. As can be seen, the Pretty Prairie city wells and nearby irrigation water samples all have nitrate-N above the drinking water limit of 10 mg/L. The monitoring wells also indicate a predominance of nitrate-N above the drinking water limit.
Figure 4. Frequency distribution of nitrate-N from irrigation, city, and monitoring wells in the vicinity of Pretty Prairie, KS. Graph shows that the majority of samples have nitrate-N greater than 10 mg/L.
A contour map of the nitrate-N values from the 12 wells sampled in August, 1999 is shown in Figure 5. This figure shows that nitrate in the vicinity of Pretty Prairie is still very high. A contour map of the area around Pretty Prairie based on data from the 1995 and current study is shown in Figure 6. Values are high in the vicinity of Pretty Prairie and lower away from the city.
Figure 5. Contour map of nitrate-nitrogen from 12 sampling points in August, 1999. Map shows nitrate-N values greater than 10 mg/L for all sites. Ground-water flow is to the north-northeast (Figure 2).
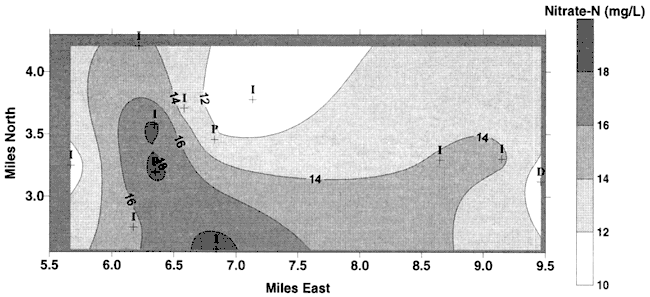
Figure 6. Contour map of nitrate-N values from 1995 study (Dealy, 1995) plus 12 samples from August, 1999. Ground-water flow is to north-northeast and does not appear to affect the nitrate distribution.
Nitrogen-15 isotope analysis is a method to assist in determining sources of nitrogen in groundwater. The isotope analysis evaluates the ratio of nitrogen-14 (what is present in the air we breathe) to the amount of nitrogen-15 present in the water or whatever is being analyzed. This ratio is compared to a standard based on the ratio of 14N/15N present in air. Comparisons of these values indicates if there is more (a positive value +) or less (negative value -) than the standard. The plus or minus sign indicates whether the sample is enriched (+) or depleted (-) in relation to the standard. Researchers have found that sources of nitrogen in groundwater fall into various categories. These are illustrated in Figure 7.
Figure 7. Range of nitrogen-15 values from multiple studies (compiled by Heaton, 1986).
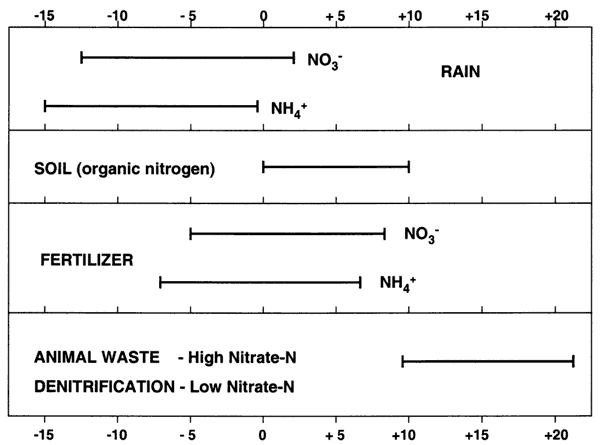
Values of δ15N that range from negative values up to +8 generally indicate a source from atmospheric nitrogen (rain), fertilizer, or soil organic nitrogen (Figure 7). The amount of nitrate measured in the sample indicates the possible sources. Rainwater has very low nitrogen content as nitrate (NO3-) or ammonium (NH4+), usually less than 1 or 2 mg/L. Fertilizer and soil organic nitrogen samples are frequently above 3 mg/L, which is considered a background level found everywhere, based on the work of researchers over many years. Generally, if fertilizer is a source, the nitrate-N concentration is above 3 mg/L and often over 10 mg/L (drinking water limit), and the δ15N signature is between +2 and +8‰.
Animal waste sources have a signature which is generally greater than +10‰. Nitrate-nitrogen concentrations are often above the drinking water limit of 10 mg/L. Another possibility for an enriched value (greater than +10) is called denitrification. This occurs when bacteria break down the nitrate-N to nitrogen gas. This process usually results in nitrate-N concentration of below 2 mg/L as well as a high δ15N value (greater than +10‰).
Nitrate-N and δ15N values for the sites sampled at Pretty Prairie are presented in Table 1. As can be seen the majority of samples are around +8‰. or below which indicates a probable fertilizer source for the nitrate-N. Figure 8 shows the δ15N values from the August sampling. Figure 8 shows the δ15N values from the study done in 1995. The δ15N values are still within the fertilizer range in August, 1999 for the Pretty Prairie area.
Table 1. Nitrate-N and δ15N for samples collected in August, 1999. *Analysis not run on sample.
| ID | NO3-N (mg/L) | δ15N ‰ | Depth of well (ft) | Well Type |
|---|---|---|---|---|
| 9JW-040 | 13.0 | 7.54 | 93 | I |
| 9JW-041 | 14.5 | * | 102 | I |
| 9JW-042 | 18.5 | * | 96 | I |
| PP-1 | 10.7 | 8.23 | 100 | I |
| PP-2 | 15.5 | 5.25 | 68 | I |
| PP-3 | 18.5 | 7.72 | 70 | P |
| PP-4 | 12.3 | 7.55 | 98 | P |
| PP-5 | 10.4 | 7.38 | 92 | P |
| PP-6 | 18.8 | 5.53 | 103 | I |
| PP-7 | 10.9 | 5.99 | 60 | D |
| PP-8 | 14.4 | 7.76 | 51 | I |
| PP-9 | 14.1 | 7.71 | 68 | I |
Figure 8. Values of δ15N ‰ for 10 sites sampled in August, 1999. Values are within the fertilizer range shown in figure 7. Rectangle represents Pretty Prairie, KS.
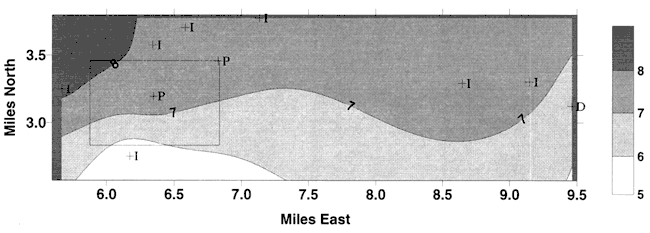
Figure 9. Values of δ15N ‰ for monitoring sites sampled in 1995 study (Dealy, 1995) Rectangle represents Pretty Prairie, KS. Values are similar to those observed in this study shown in Figure 8.
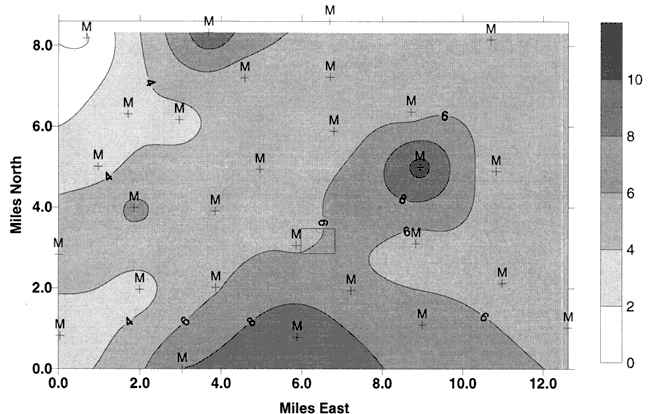
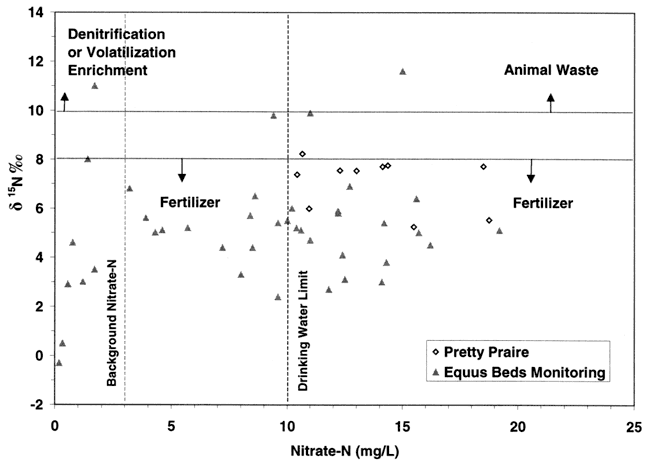
Figure 10 shows another plot of the wells from this study (Pretty Prairie) and the Equus Beds samples from the 1995 study (Dealy, 1995). This graph illustrates the potential sources for nitrate in relation to the nitrogen-15 signature. The line at δ15N of +8 ‰ and below indicates a fertilizer source. The line at +10 ‰ indicates human and animal waste sources. The vertical line at 3 mg/L nitrate-N indicates the background level for natural nitrate-N occurrence, and 10 mg/L nitrate-N indicate the drinking water limit. The majority of samples from both the current and the 1995 study (Dealy, 1995) plot within the fertilizer source range.
Figure 10. Graph shows location of δ15N values for this study (Pretty Prairie) and the 1995 study (Equus Beds wells) in relation to nitrate-nitrogen concentration. Most points plot in the fertilizer source range.
The area around Pretty Prairie has a vertical topographic relief difference of approximately 30 ft across the 3 mile study area. The presence of intermittent streams indicates a lack of perennial surface flow and is an indicator that water recharged at the surface either is evaporated or flows downward to the ground water. The majority of nitrate-nitrogen concentrations in ground water are above the drinking water limit of 10 mg/L and have been for some time. Use of nitrogen-15 indicates a predominantly fertilizer source for the nitrate. A previous study in the area also found a similar source with the additional chemical evidence of the presence of atrazine and atrazine degradation products in the ground water.
Bayne, C.K., 1956, Geology and ground-water resources of Reno County, Kansas: Kansas Geological Survey, Bulletin 120, 130 p. [available online]
Dealy, Michael T., 1995, Investigation of nitrate-nitrogen concentrations in the Equus Beds aquifer, southeast Reno County, Kansas: Equus Beds Groundwater Management District # 2, Halstead, KS, Water Quality Investigation 95-01, 148 p.
Heaton, T. H. E., 1986, Isotopic studies of nitrogen pollution in the hydrosphere and atmosphere: a review: Chemical Geology, vol. 59, p. 87-102.
Complete water chemistry analyses from sites near Pretty Prairie, KS.
| ID | Location TRS | Field Sp. C. |
Lab pH |
Ca | Mg | Na | K | Sr | HCO3 | SO4 | Cl | F | NO3-N | TDS calc. |
B (ppb) |
δ15N ‰ |
Sample Date |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| I | 26-06W-18ACA | 354 | 6.7 | 39.6 | 6.3 | 19.4 | 1.2 | 0.18 | 117 | 21.1 | 7.7 | 0.21 | 13.0 | 210.6 | 28 | 7.54 | 7/5/1999 |
| I | 26-06W-07CDB | 446 | 6.8 | 52.7 | 9 | 22.2 | 1.3 | 0.25 | 160 | 23 | 10.3 | 0.2 | 14.5 | 262.0 | 27 | 7/5/1999 | |
| I | 26-06W-18BDD | 378 | 6.6 | 44.6 | 7.2 | 15 | 1.1 | 0.21 | 95.1 | 21.5 | 9.7 | 0.16 | 18.5 | 228.0 | 20 | 7/5/1999 | |
| PP-1 | 26-07-13D | 365 | 6.7 | 38.5 | 6.8 | 20.7 | 1.0 | 0.19 | 123 | 23.9 | 10.0 | 0.25 | 10.7 | 574.1 | 27 | 8.23 | 8/5/1999 |
| PP-2 | 26-06-19B | 345 | 6.6 | 36.0 | 6.1 | 18.1 | 1.2 | 0.17 | 78.9 | 23.8 | 10.3 | 0.25 | 15.5 | 548.3 | 29 | 5.25 | 8/5/1999 |
| PP-3 | 26-06-18CDA | 440 | 7.0 | 46.6 | 7.8 | 22.1 | 1.3 | 0.22 | 122 | 23.6 | 11.2 | 0.24 | 18.5 | 695.0 | 40 | 7.72 | 8/5/1999 |
| PP-4 | 26-06-18DAA | 360 | 6.6 | 38.1 | 5.9 | 19.8 | 1.2 | 0.17 | 117 | 22.1 | 6.4 | 0.20 | 12.3 | 565.8 | 30 | 7.55 | 8/5/1999 |
| PP-5 | 26-06-17B | 340 | 6.6 | 36.5 | 6.5 | 18.5 | 1.3 | 0.18 | 125 | 16.6 | 4.9 | 0.23 | 10.4 | 532.3 | 36 | 7.38 | 8/5/1999 |
| PP-6 | 26-06-19ADD | 390 | 6.8 | 41.1 | 7.0 | 20.3 | 1.3 | 0.20 | 96.1 | 27.0 | 7.4 | 0.24 | 18.8 | 624.8 | 26 | 5.53 | 8/5/1999 |
| PP-7 | 26-06-15DCC | 320 | 6.6 | 32.5 | 6.2 | 18.2 | 1.3 | 0.18 | 101 | 19.7 | 5.1 | 0.23 | 10.9 | 501.6 | 28 | 5.99 | 8/5/1999 |
| PP-8 | 26-06-15C | 360 | 6.9 | 35.9 | 7.3 | 182 | 1.3 | 0.21 | 96.6 | 22.8 | 9.6 | 0.21 | 14.4 | 566.6 | 30 | 7.76 | 8/5/1999 |
| PP-9 | 26-06-16D | 370 | 6.8 | 38.0 | 8.0 | 19.2 | 1.3 | 0.23 | 118 | 21 8 | 5.2 | 0.25 | 14.1 | 584.7 | 35 | 7.71 | 8/5/1999 |
Units of Sp. C. (Specific Conductance) in μmhos/cm.
Units of all chemical parameters (except B and 15N) in mg/L
Ca = Calcium; Mg = Magnesium; Na = Sodium; K= Potassium; Sr = Strontium; HCO3 = Bicarbonate;
SO4 = Sulfate; Cl Chloride; F = Fluoride; NO3-N = Nitrate-nitrogen;
TDS (calc.) = Total Dissolved Solids calculated from chemical analyses; B = Boron in ppb;
δ15N ‰ = nitrogen-15 isotope values in per mil (ppt).
Cross-section of well logs from sampled wells. Sand and gravel predominate in the unsaturated and saturated zone profile. Silt and clay zones are not generally continuous.
Kansas Geological Survey, Geohydrology
Placed online Nov. 14, 2007
Comments to webadmin@kgs.ku.edu
The URL for this page is http://www.kgs.ku.edu/Hydro/Publications/1999/OFR99_44/index.html