
 |
Original published in D.F. Merriam, ed., 1964, Symposium on cyclic sedimentation: Kansas Geological Survey, Bulletin 169, pp. 191-197 | |
The University of Michigan, Ann Arbor, Michigan
Evaporites are characterized in a vertical sequence by cycles of rock types, such as dolomite-anhydritehalite-anhydrite-dolomite. Associated with the rock sequences are textures, peculiar to certain rock types, which also occur in cyclic sequences. In the Devonian Lucas Formation of Michigan the textures associated with anhydrite-dolomite rocks are decussate anhydrite, nodular anhydrite, reticulated anhydrite, and massive anhydrite. The textures can be related in a general way to physicochemical conditions of mineral precipitation; thus, they are clues to changing conditions of evaporite deposition.
Cycles in evaporitic rocks are most evident in the repetitive sequences of the major evaporitic rock types-dolomite-anhydrite-dolomite, dolomite-anhydrite-halite-anhydrite-dolomite, etc. Within these cycles are other smaller scale repetitive sequences, such as a laminae or "varves" in anhydrite-calcite rocks described by Udden (1924). The textures of evaporitic rocks show a similar type of cyclicity that appears to be related to the salinity and chemical composition of the brine from which the minerals are precipitating by evaporation of the water and concentration of the brine. Such textural cycles are well known from the Permian evaporites of Germany (Richter-Bernberg, 1955; Hoyningen-Huene, 1957) and from the Miocene sulfate rocks of Sicily (Ogniben, 1955, 1957), which Dr. W. T. Holser brought to our attention. Dellwig (1955) described cyclic textures in rock salt from the Silurian Salina Formation of Michigan. In this paper cyclic textures are described from the Devonian Lucas Formation of Michigan.
Acknowledgments--This work was supported by the University of Michigan Institute of Science and Technology, Project 21. We appreciate the aid and criticism given by our colleagues, especially Frank Moser, Donald Ehman, David Hixon of the University of Michigan and William Mantec of the Michigan Geological Survey.
Evaporitic minerals precipitate from a supersaline brine in which the salts are concentrated by net water evaporation loss. Mineral deposition progresses from carbonate minerals to sulfate minerals to the more soluble salts-halite, polyhalite, and bittern salts, as illustrated in Table 1.
TABLE 1--Theoretical progression of evaporitic salts above 30°C. Modified after Stewart, 1963, p. Y23.
| Polyhalite and halite, or glauberite and halite |
| Anhydrite and halite |
| Anhydrite |
| Limestone and/or dolomite |
Mineral precipitation is very sensitive to the degree of concentration of the supersaline brine and to climatic changes in temperature, because temperature and humidity greatly affect evaporation rates. When the water lost by evaporation exceeds that supplied by rainfall, rivers, and oceans, there is a progressive increase in salinity of the evaporitic brine.
The cyclic nature of these deposits originates by periodic refreshening of the concentrated brine owing to changes in rainfall, influx of seawater into the evaporitic basin, terrestrial runoff, or evaporation. Fundamentally, cyclicity of evaporitic deposits results from periodic climatic changes. These may be due to solar activity (Anderson, 1963, p. 891) or to other climatological factors. Anderson noted cycles having periods of 12 to 16 years, 80-90 years, and 180 years in his study of the evaporitic varves of the Jurassic Todilto Formation in New Mexico (Anderson, 1963, p. 887-892). Periods of this order might account for some of the evaporitic rock cycles observed in the Lucas Formation of Michigan. The consecutive precipitation of saline minerals (Table 1), corresponding to increasing concentration of the evaporitic brine, is often followed by the precipitation of less saline minerals in reverse order of solubility. Interruptions of a particular sequential pattern, especially omissions of one or more components, are common.
The Middle Devonian Lucas Formation represents a major evaporitic sequence of dolomite, anhydrite, and halite in Michigan (Fig. 1). The thicknesses of the major evaporitic cycles range from 3 to 150 feet; most are between 5 and 30 feet. Rock textures associated with these rocks are also cyclic in vertical distribution. The textures can be related in a general way to physicochemical environments of evaporite rock deposition (Table 2) by the proportions of anhydrite and halite in the associated rocks which are indicative of the salinity of the evaporitic brine (Usiglio, 1849; Clarke, 1924, p. 220; Briggs, 1957; p. 47; see, Fig. 2A, 2B, 2C, 3A).
Figure 1--Index map of Michigan and adjacent areas.
Table 2--Sequence of rock types in a Lucas evaporitic unit.
| Type of rock | Approximate anhydrite percentage in rock |
Approximate halite percentage in rock |
Estimated salinity of evaporate brine ‰ |
|---|---|---|---|
| Rocksalt, (halite) | 0 | 100 | greater than 340 |
| Salt with abundant anhydrite crystals | 10±5 | 90±5 | 300 to 340 |
| Salt interbedded with anhydrite | 50±20 | 275 to 300 | |
| Massive anhydrite | 100 | 0 | 200 to 275 |
| Anhydrite, mosaic | 95±5 | 0 | 200 to 275 |
| Anhydrite with thin beds of dolomite, 1-6" | 75±10 | 0 | 150 to 200 |
| Anhydrite, reticulated | 60±20 | 0 | 150 to 200 |
| Admixed anhydrite-dolomite | 50±20 | 0 | 150 to 200 |
| Dolomite with nodular anhydrite | 10±5 | 0 | less than 150 |
| Dolomite with crystals of anhydrite | 5±5 | 0 | less than 150 |
| Dolomite | 0 | 0 | less than 150 |
Figure 2--A, Recrystallized salt and anhydrite in core in Pure Reinhardt No. 1, depth 3649 feet, X 0.6. B, Massive anhydrite in core in Sun St. Mancelona No. A-1, depth 3626 feet, X 1.4. C, Nodular anhydrite and laminated dolomite in core in Pure Joseph No. 6, depth 4170 feet, X 1.0. D, Reticulated anhydrite in core in Pure Reinhardt No. 1, depth 3737 feet, X 1.0.
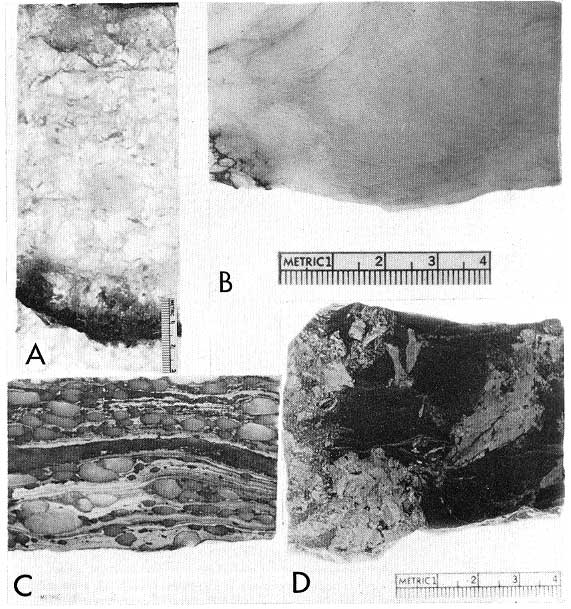
Figure 3--A, Decussate anhydrite in photomicrograph of core in Pure Joseph No. 6, depth 4265 feet, X 32, crossed nicols, lathlike crystals of anhydrite cut across mosaic of celestite crystals showing partial replacement by small colloform masses of chalcedony. B, Flowage structure displayed in microfeltic anhydrite, photomicrograph of core in Sun St. Mancelona No. A-1, depth 3770 feet, X 63, crossed nicols.
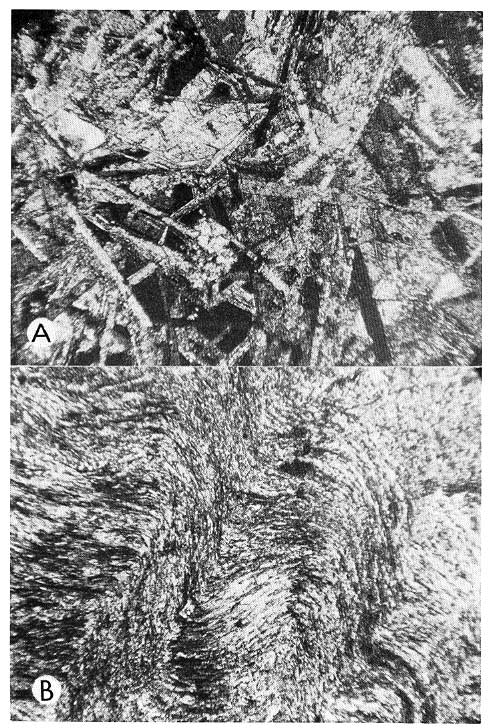
Evaporitic rock textures are related also to other factors, such as density flowage, slumping, and microbrecciation while the deposit is still in a semifluid or thixotropic state (Riley and Byrne, 1961; see, Fig. 2D, 3B).
A typical Lucas evaporitic sequence is dolomite, anhydrite, and halite (Fig. 4). During deposition of the Lucas Formation, the brine concentration did not always reach the halite stage before dilution occurred. Part of this variation in any one core may be due to the lateral variation in brine concentration (salinity facies) over the basin, the distribution of which may have varied from time to time during deposition of the Lucas evaporites. Thick anhydritic deposits may be related to long periods of stability of the basin brine and to the stage of concentration of the brine. As examples of the concentration effect, Usiglio's data shows that a rate of sulfate mineral precipitation (mg per ml water evaporated) of 15.8 at salinity 222-240 ‰, but only 10.2 at 157.222 ‰ and 8.4 at 274 ‰ (Briggs, 1957, 1958; see, Fig. 5).
Figure 4--Halite and anhydrite cycles in Lucas Formation (from Carter Schmidt No. 1, Grand Traverse County, Michigan).
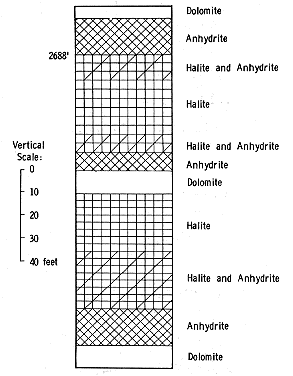
The reverse sequence with progressive deposition of less soluble salts following halite precipitation probably is largely the result of a higher rate of influx of seawater into the basin (Fig. 4). Terrestrial runoff was not an important factor in refreshening or disrupting the evaporation of the basin brine during deposition of the Lucas Formation inasmuch as clay and shale are insignificant components of the rocks.
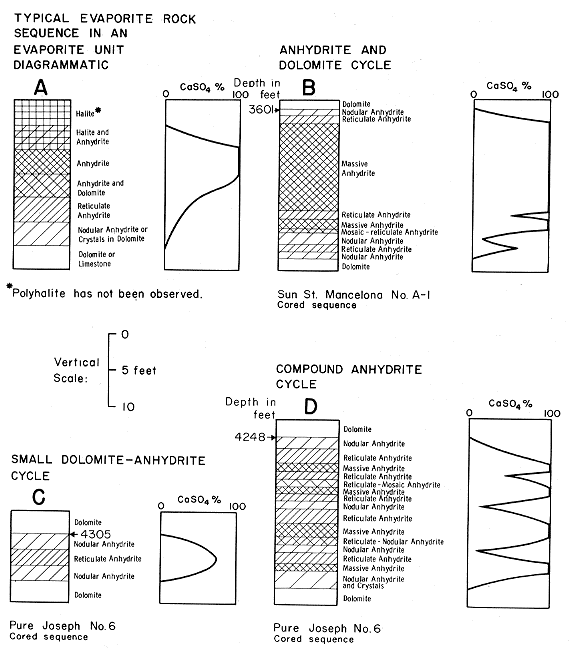
Figure 5--Evaporite cycles showing anhydritic textures in Lucas Formation.
Evaporitic rocks result from the deposition of salts precipitated by evaporation of water from concentrated natural brines in which there is a delicate balance among evaporation, rainfall, influx of new sea water, and terrestrial runoff of rivers into the evaporitic basin. At any one time during deposition of the rocks a dynamic equilibrium or steady state exists among these variables. Over a period of time the equilibrium shifts, changing the composition of the brine and the character of mineral precipitation. Such shifts in equilibrium are normally gradual and repetitive, resulting in repetitive sequences in evaporitic rocks and evaporitic rock cycles.
Asssociated with the evaporitic rock cycles are cycles in evaporitic rock textures, produced under specific conditions of brine concentration and other physicochemical conditions. Although the specific conditions producing a particular texture are but approximately known, the sequences of textures can be related to sequential changes in the environments of evaporitic rock deposition.
Anderson, R. Y., 1963, Harmonic analysis of varve time series: Jour. Geoph. Research, v. 68, no. 3, p. 877-893.
Briggs, L. I., 1957, Quantitative aspects of evaporite deposition: Mich. Acad. Sci. Papers, v. 42, p. 115-123.
Briggs, L. I., 1958, Evaporate facies: Jour. Sed. Pet., v. 28, p. 46-56.
Clarke, F. W., 1924, The data of geochemistry: U. S. Geol. Survey Bull. 770, 841 p.
Dellwig, L. F., 1955, Origin of the Salina salt of Michigan: Jour. Sed. Pet., v. 25, p. 83-110.
Hoyningen-Huene, E. v., 1957, Die Texturen der subsalinaren Anhydrit im Harzvorland und ihre stratigraphische und fazielle Bedeutung: Geologie, Beiheft nr. 18.
Ogniben, L., 1955, Inverse graded bedding in primary gypsum of chemical deposition: Jour. Sed. Pet., v. 25, p. 273-281.
Ogniben, L., 1957, Secondary gypsum of the sulfur series, Sicily, and the so-called integration: Jour. Sed. Pet., v. 27, p. 64-79.
Richter-Bernberg, G. v., 1955, Über salinare Sedimentation: Deutsche Geol. Gesell. Zeitschr., v. 105, p. 593-645.
Riley, C. M., and Byrne, J. V., 1961, Genesis of primary structures in anhydrite: Jour. Sed. Pet., v. 31, p. 553-559.
Stewart, F. H., 1963, Marine evaporites, in Data of geochemistry, 6th edition: U. S. Geol. Survey Prof. Paper 440-Y, 52 p.
Udden, J. A., 1924, Laminated anhydrite in Texas: Geol. Soc. America Bull., v. 35, p. 347.354.
Usiglio, J., 1849, Analyse de I'eau de la Méditerranée sur les côtes de France: Annalen der Chemie, v. 27, p. 92.107, 172-191.